
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Professional technology integration to support the entire R&D process
Home>
Resources>
Learning>
Articles>
Targeting 5-HTR2: A Versatile Receptor from the Nervous System to Metabolic Diseases and Its Drug Screening Models
Targeting 5-HTR2: A Versatile Receptor from the Nervous System to Metabolic Diseases and Its Drug Screening Models
In the fields of neuroscience and drug discovery, serotonin (5-HT) is undoubtedly one of the most extensively studied signaling molecules. However, recent research has revealed that the functions of 5-HT extend far beyond the regulation of mood, behavior, and cognition. As a multifunctional signaling mediator, 5-HT also plays a critical role in pathological states such as tumorigenesis, inflammatory responses, and pathogen infections. Among these, the 5-HT2 receptor family (5-HTR2) has become a vital therapeutic target due to its broad tissue distribution and diverse physiological functions. Leveraging its mature GPCR drug screening platform, Reqbio has developed a series of cell models covering the 5-HTR2A, 5-HTR2B, and 5-HTR2C subtypes. These models are designed to provide researchers worldwide with high-efficiency and reliable tools for early-stage R&D.
I. The 5-HTR2 Family: Diversity in Structure and Function
The 5-HTR2 family comprises three receptor subtypes: 5-HTR2A, 5-HTR2B, and 5-HTR2C, which share 42–51% amino acid sequence homology. All three receptors primarily couple to Gq/G11 proteins. Upon activation, they trigger a signaling pathway mediated by phospholipase C (PLC), leading to the increased production of inositol trisphosphate (IP3) and diacylglycerol (DAG), which subsequently elevates intracellular Ca²⁺levels. Furthermore, these receptors can activate downstream signaling pathways such as PI3K/Akt and ERK, regulating biological processes including cell proliferation, differentiation, survival, and migration.Notably, the 5-HTR2C receptor possesses a unique RNA editing mechanism that generates multiple isoforms with distinct functions. These edited isoforms may exhibit different signal transduction efficiencies and pathway biases, presenting both opportunities and challenges for drug discovery. Psychotropic drugs can modulate this RNA editing to alter 5-HTR2C function in a brain-region-specific manner, thereby enabling precision medicine approaches.
II. 5-HTR2A: A Key Regulator of Cognition and Emotion
5-HTR2A is widely distributed throughout the central nervous system (CNS) and peripheral tissues. In the brain, it is primarily enriched in the neocortex, hippocampus, and amygdala—regions closely associated with cognition, emotion, and neuroplasticity. Specifically, on the apical dendrites of layer V pyramidal neurons in the cerebral cortex, 5-HTR2A finely tunes working memory, attention, and executive functions by modulating glutamate release and the activity of GABAergic interneurons.Complexity of Signal Transduction: 5-HTR2A can simultaneously activate two distinct signaling pathways: the Gq protein pathway and the β-arrestin pathway, each mediating different physiological effects. The Gq pathway primarily initiates the classical PLC/IP3/DAG cascade, while β-arrestin is involved in receptor internalization, desensitization, and unique intracellular signaling. This biased signaling mechanism makes it possible to develop highly selective agonists or antagonists, yet significantly increases the complexity of drug discovery.Currently, 5-HTR2A antagonists (such as Clozapine) and inverse agonists are widely used to treat schizophrenia, cognitive impairment, and migraines. However, due to the intricate nature of these receptor signaling pathways, developing safe and effective 5-HTR2A agonists remains a formidable challenge.
Ⅲ.5-HTR2B: A Potential Target for Cardiovascular Disease and Fibrosis
Since its first characterization in 1992, research on 5-HTR2B has made significant strides. It plays a vital regulatory role across the cardiovascular system, fibrotic diseases, cancer, the gastrointestinal tract, and the nervous system. The binding modes of 5-HTR2B with various ligands have provided unique insights into the mechanisms of receptor activation and biased signaling.
Currently, several selective 5-HTR2B antagonists (such as RS-127445 and PRX-08066) have been developed for the treatment of pulmonary arterial hypertension (PAH), valvular heart disease, and fibrotic disorders. Furthermore, the role of 5-HTR2B within the tumor microenvironment is gaining increasing attention, positioning it as a promising novel target for cancer therapy.
Ⅳ.5-HTR2C: The Star Target for Anti-Obesity Medications
5-HTR2C was among the first serotonin receptors to be sequenced and cloned, with its gene localized on the X chromosome. Unlike 5-HTR2A and 5-HTR2B, the 5-HTR2C gene contains three introns and encodes a protein characterized by seven transmembrane (7TM) domains. It is widely expressed throughout the central nervous system (CNS), particularly in key brain regions responsible for regulating energy metabolism, including the ventral tegmental area (VTA), the arcuate nucleus (ARC), and the paraventricular nucleus of the hypothalamus (PVN).
Mechanism of 5-HTR2C in Energy Metabolism:
Upon the binding of serotonin to 5-HTR2C, the Gq/G11 protein is activated, initiating the PLC/IP3/DAG/PKC signaling cascade. This, in turn, triggers the ERK pathway, promoting the phosphorylation of c-Fos and the synthesis of POMC (Pro-opiomelanocortin). POMC is subsequently processed into α-MSH, which induces the activation of neurons in the paraventricular nucleus (PVN) by activating melanocortin 4 receptors (MC4R). This biological sequence ultimately generates a feeling of satiety and inhibits food intake.
History and Insights from Drug Discovery:
Several drugs targeting 5-HT signaling have historically been used as appetite suppressants, including Sibutramine, mCPP, and Fenfluramine. Research has demonstrated that the anorectic effects of mCPP are completely abolished in 5-HTR2C knockout mice, confirming the central role of 5-HTR2C in d-fenfluramine-induced satiety. However, Fenfluramine was eventually withdrawn from clinical use due to severe side effects—namely valvular heart disease (VHD) and pulmonary arterial hypertension (PAH)—which were primarily mediated by 5-HTR2B. This historical lesson underscores the critical importance of subtype selectivity in drug development and has positioned 5-HTR2C as one of the most promising targets for next-generation anti-obesity medications.
Ⅴ.Accelerating 5-HTR2 Drug R&D: Reqbio’s One-Stop Cell Models
In the field of GPCR-targeted drug discovery, reliable functional screening models are the cornerstone for identifying lead compounds and optimizing drug candidates. Leveraging years of expertise, Reqbio has successfully developed and validated a comprehensive series of cell models covering all three subtypes of the 5-HTR2 family. These models are designed to meet diverse research requirements across various screening stages.
Catalog: Reqbio 5-HTR2 Series Cell Models
|
Cell Model Name |
Catalog No. |
Core Application |
|
HEK293 Human 5-HTR2A Cell Line |
RQP71555 |
Agonist/Antagonist Screening and Signaling Pathway Research |
|
HEK293 Human 5-HTR2B Cell Line |
RQP71582 |
Agonist/Antagonist Screening and Cardiovascular Drug Evaluation |
|
HEK293 Human 5-HTR2C(no edited) Receptor Cell |
RQP71598 |
Agonist/Antagonist Screening and RNA Editing Function Research |
|
HEK293 Human 5-HTR2C(edited) Receptor Cell |
RQP71583 |
Agonist/Antagonist Screening and Functional Research of Edited Isoforms |
|
CHO-K1 Human 5-HTR2B Cell Line |
RQP71575 |
CHO-K1 Agonist/Antagonist Screening and High-Throughput Screening (HTS) |
|
CHO-K1 Human 5-HTR2C(no edited) Receptor Cell |
RQP71584 |
Agonist/Antagonist Screening and High-Throughput Screening (HTS) |
|
CHO-K1 Human 5-HTR2C (edited) Cell Line |
RQP71574 |
Agonist/Antagonist Screening and High-Throughput Screening (HTS) |
Product Validation Data Display:
1.Functional Validation of HEK293 Human 5-HTR2A Cell Line(RQP71555)
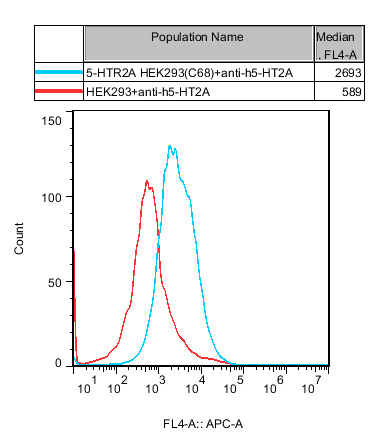
Figure 1. Recombinant 5-HTR2A HEK293 stably expressing 5-HTR2A.
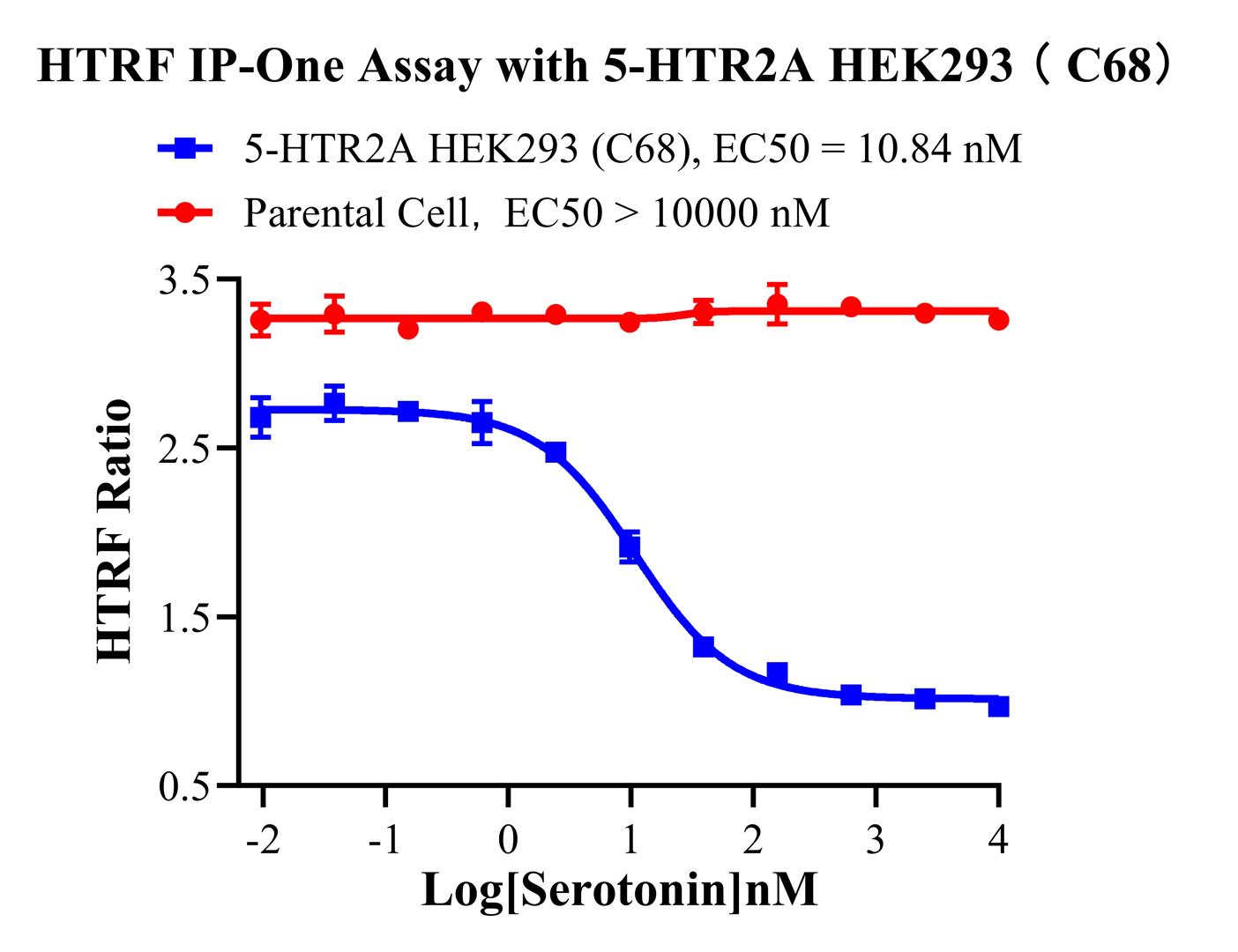
Figure 2. HTRF IP-One Assay with 5-HTR2A HEK293(C68).
2.Functional Validation of HEK293 Human 5-HTR2B Cell Line(RQP71582)
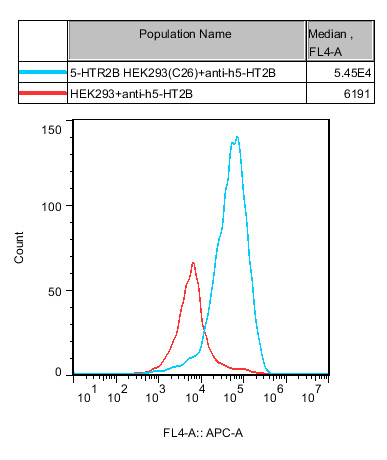
Figure 3. Recombinant 5-HTR2B HEK293 stably expressing 5-HTR2B.
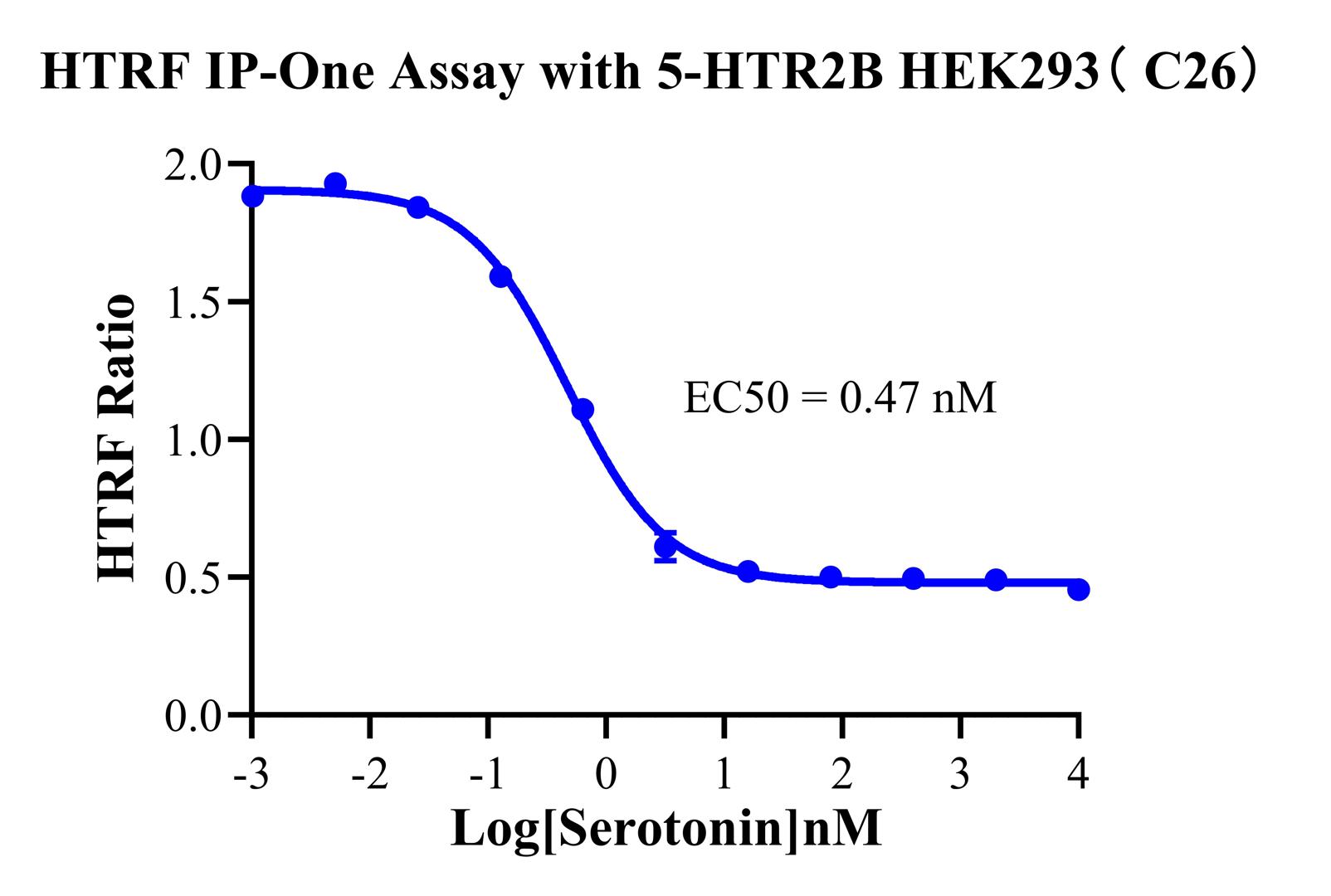
Figure 4. HTRF IP-One Assay with 5-HTR2B HEK293( C26).
3.Functional Validation of HEK293 Human 5-HTR2C(no edited) Receptor Cell(RQP71598)
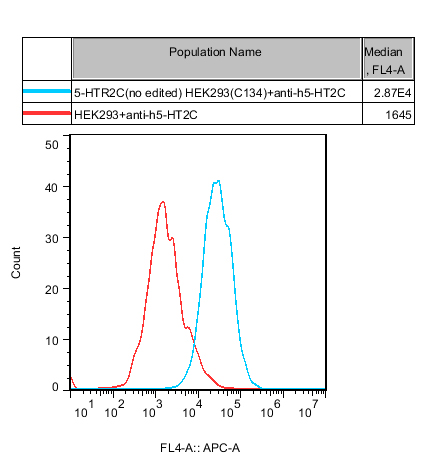
Figure 5. Recombinant 5-HTR2C(no edited) HEK293 stably expressing 5-HTR2C(no edited).
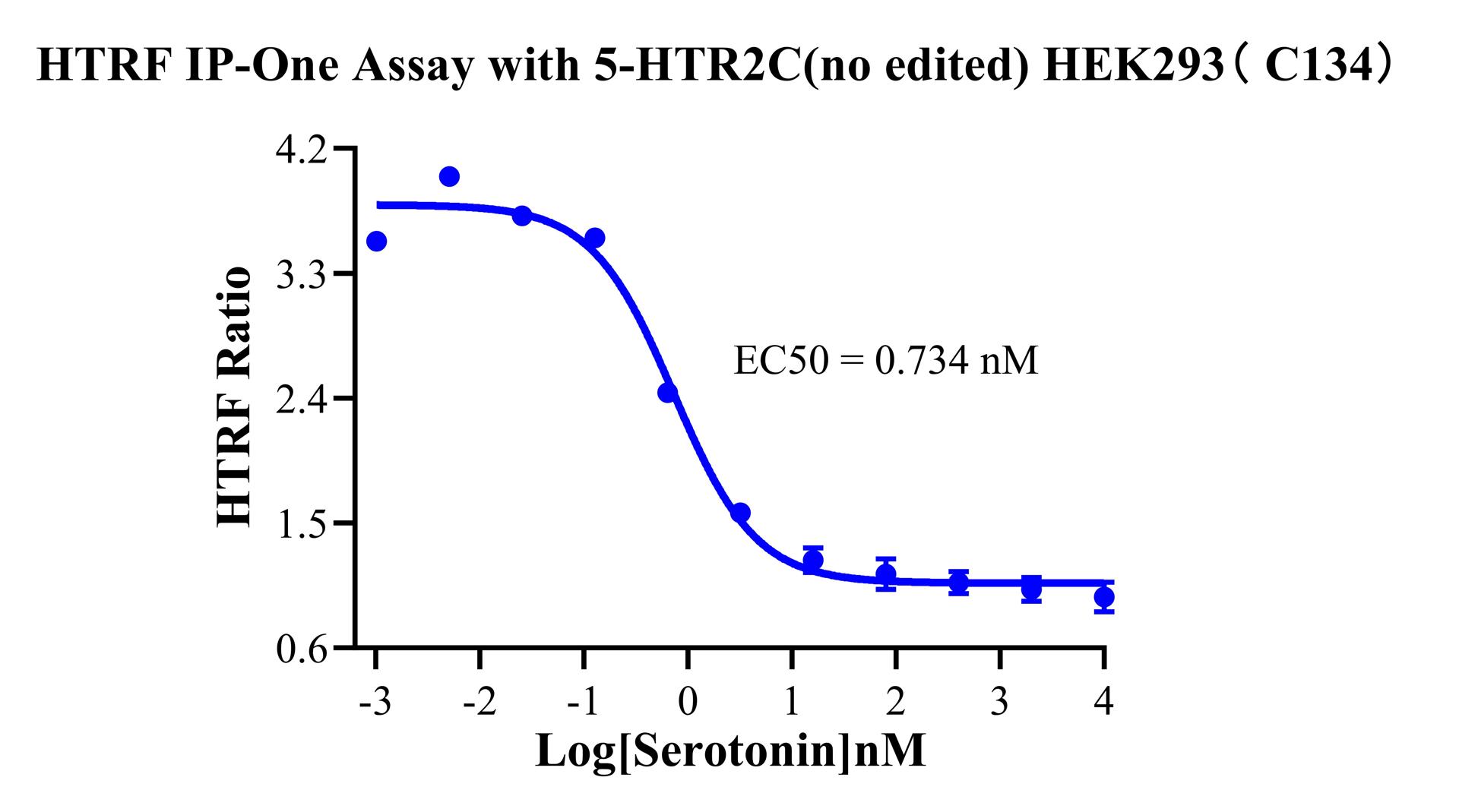
Figure 6. HTRF IP-One Assay with 5-HTR2C(no edited) HEK293 HEK293 (C134).
Product Core Advantages:
High Sensitivity and Stability: Rigorous functional validation ensures excellent consistency and reproducibility of experimental data.
Diverse Detection Platforms: Support for multiple assay modes, including $\beta$-arrestin recruitment, cAMP inhibition, and calcium flux, meeting diverse screening requirements.
Ready-to-Use Products: Streamlined experimental workflows shorten R&D cycles, helping customers accelerate early-stage drug discovery.
Ⅴ.Conclusion
As our understanding of the tumor microenvironment deepens, targeting the CCL2-CCR2 axis is transitioning from concept to clinical reality. Whether you are developing small molecule inhibitors, neutralizing antibodies, or novel ADCs, reliable screening models are the cornerstone of success. Reqbio remains committed to focusing on frontier targets in GPCRs and immuno-oncology, providing high-quality biological reagents and cell models to empower global innovative drug discovery.
news recommendation
NFAT Signaling Pathway: A Critical Target for Drug R&D and Cell Models
TSHR Cell Model for Thyroid Cancer Drug Screening | GPCR Assay Platform-Reqbio
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore



