
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Professional technology integration to support the entire R&D process
Home>
Resources>
Learning>
Articles>
Targeting C3aR: A New Chapter in the Complement System -A Key Target from Inflammatory Regulation to Cancer Immunotherapy
Targeting C3aR: A New Chapter in the Complement System -A Key Target from Inflammatory Regulation to Cancer Immunotherapy
Main Text: Analysis of C3aR Signaling Mechanisms: A Core Target from Basic Research to Drug Development
As a central component of the innate immune system, the aberrant activation of the complement system cascade is closely linked to the onset and progression of various diseases, including inflammation, cancer, and neurodegenerative disorders. The Complement C3a Receptor (C3aR), a key node within the complement system and a member of the G Protein-Coupled Receptor (GPCR) family, has become a focal point of research in the biopharmaceutical field. Precise regulation of its signal transduction pathways has emerged as a vital target for novel drug R&D, demonstrating immense potential particularly in the field of cancer immunotherapy.
I.Core Mechanisms of C3aR Signaling
The complement cascade can be activated through three pathways: the classical, lectin, and alternative pathways. All three trigger early reactions that generate C3 convertase, which cleaves the central complement component C3 into C3a and C3b. Within this process, C3b acts as an opsonin to activate the lytic pathway, while C3a acts as a soluble factor. Through autocrine and paracrine signaling, C3a specifically binds to its receptor, C3aR, to regulate innate immune cells and participate in the modulation of both local and systemic inflammatory responses.
C3aR belongs to the GPCR family and possesses the characteristic seven-transmembrane (7TM) domains. The nature of its coupling with G proteins has long been a subject of research debate; while some literature indicates activation of the Gαi family, other studies have suggested the involvement of Gαs and Gαq in its signal transduction.Latest research has clarified that C3aR primarily couples with Gαi/o/z subtypes. Its core function is to inhibit cAMP accumulation and mediate calcium influx—an increase in intracellular calcium concentration is the most typical downstream response following C3aR activation. Furthermore, several molecular pathways, including GSK3β, Wnt, ERK, JAK/STAT, and HIF1α, serve as downstream regulatory pathways for C3aR. These pathways participate in the modulation of various physiological and pathological processes such as inflammation and metabolism, providing multi-dimensional targets for the development of C3aR-targeted drugs.
II. The Remarkable Encounter of Two "Ligands": C3a and TLQP-21
C3a is the most classic ligand for C3aR, consisting of 77 amino acids, with its C-terminal region being critical for agonistic activity. However, once formed, C3a is rapidly converted by carboxypeptidases into C3a-desArg, which loses its ability to bind to C3aR, resulting in an extremely short half-life.
Breakthrough research in recent years has discovered that TLQP-21—a secretory peptide derived from the neuropeptide VGF (named after its first four N-terminal amino acids: Thr-Leu-Gln-Pro)—is a high-affinity ligand for C3aR. While TLQP-21 exists in a disordered coil conformation in its free state, it transitions into an ordered conformation upon binding with C3aR. This binding can be specifically blocked by C3aR antagonists and is abolished in C3ar1 knockout cells.
|
Characteristic |
C3a |
TLQP-21 |
| Source | Complement C3 cleavage product | Neuropeptide VGF cleavage product |
| Half-life | Extremely short (Rapid degradation) | Approx. 110 minutes (Stable) |
| Binding Affinity (EC₅₀) |
Mouse: ~3x higher than TLQP-21 |
Lower affinity, but more stable |
| Conformation | Ordered in activated state | Disordered in free state; Ordered upon binding |
This discovery has opened new doors for C3aR target research: as a long-acting and stable ligand, TLQP-21 provides a more practical tool molecule for drug screening. It also suggests that C3aR may possess regulatory mechanisms involving biased signaling.
III. Signal Regulation of C3aR: From Activation to Desensitization
In its resting state, C3aR is coupled with Gαi proteins. Upon the binding of C3a or TLQP-21, GDP is exchanged for GTP, causing the dissociation of Gαi from the Gβγ complex, which initiates downstream signaling: inhibition of adenylate cyclase → decreased cAMP levels → increased calcium flux. Subsequently, transcription factors STAT3 and HIF-1α are activated, regulating the expression of genes associated with inflammation, lipid/glucose metabolism, and OXPHOS (oxidative phosphorylation) inhibition.
To terminate the signal, GRK2/3 kinases phosphorylate the cytoplasmic loops and C-terminal tail of C3aR, facilitating the recognition and recruitment of β-arrestin. The binding of β-arrestin triggers the internalization of the receptor complex, which is then sorted within endosomes: one portion is recycled back to the cell surface via recycling endosomes, while the other is directed to lysosomes for degradation. This sophisticated regulatory mechanism provides the theoretical foundation for developing biased ligands—compounds that selectively activate either the G-protein pathway or the β-arrestin pathway.
IV. C3aR-Targeted Drug R&D: From Inflammation to Cancer Immunotherapy
The activation of the C3a-C3aR axis does not only regulate bacterial infections and innate immune responses; in recent years, it has been identified as a new class of immune checkpoint receptors. Within the tumor microenvironment (TME), C3aR signaling can influence the functions of tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs), thereby participating in immune evasion. Consequently, C3aR antagonists or agonists hold promise as novel strategies for cancer immunotherapy.
Currently, global drug development targeting C3aR is primarily in the preclinical or early stages, with modalities including small molecule chemicals and monoclonal antibodies (mAbs). As our understanding of C3aR structural biology and biased signaling deepens, this target is poised for a breakthrough.
In GPCR drug discovery, selecting a cell model that is stable, sensitive, and functionally validated is critical to success. ReqBio has developed two core cell models for the C3aR target to cover different detection platforms and screening requirements.
ReqBio’s C3aR Series Cell Models:
|
Cell Name |
Cat. No. |
Host Cell |
Detection Mode |
Core Applications |
| CHO-K1 Human C3aR Cell Line | RQP71375 | CHO-K1 | HTRF cAMP Assay | Gαi signaling pathway activity evaluation; Agonist/Antagonist screening |
| CHO-K1 Human C3aR β-Arrestin Cell Line | RQP71386 | CHO-K1 | β-Arrestin Recruitment Assay | Biased ligand screening; Desensitization and internalization research; High-throughput screening (HTS) |
Product Validation Data Display:
1.CHO-K1 Human C3aR Cell Line(RQP71375)
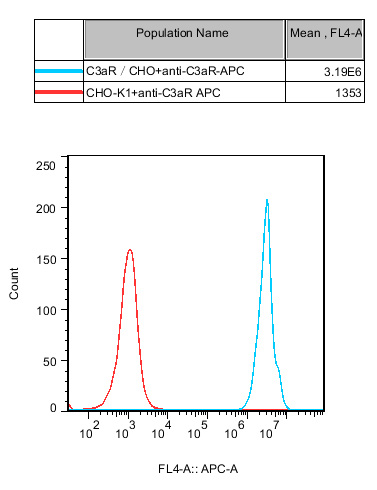
Figure 1. Flow cytometry or Western Blot confirms the stable and high-level expression of C3aR in CHO cells, ensuring the reliability of the screening system.
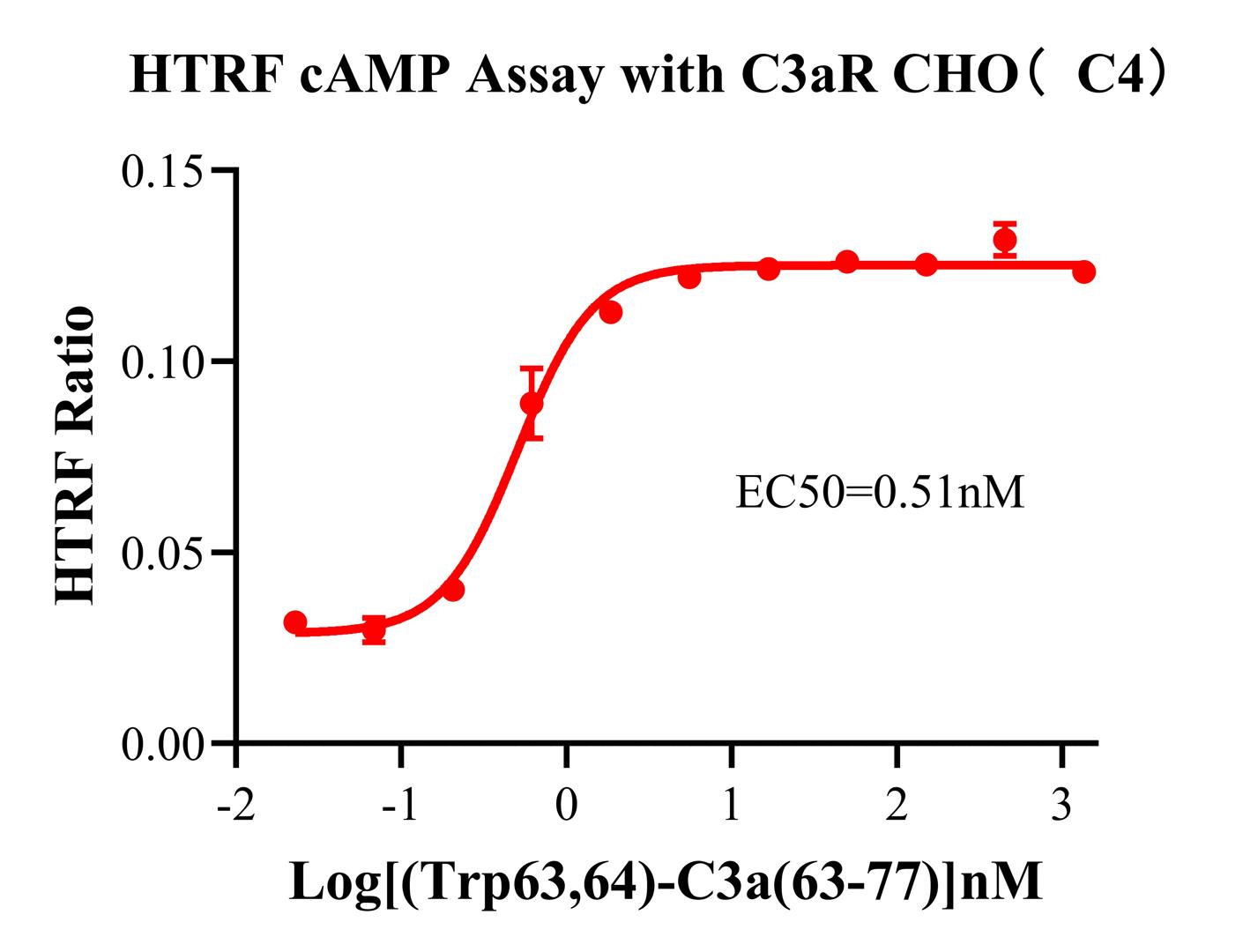
Figure 2.Evaluation of the dose-response effect of C3aR agonists on cAMP inhibition using the HTRF (Homogeneous Time-Resolved Fluorescence) cAMP detection platform. This assay offers advantages such as high sensitivity, high signal-to-noise ratio, and ease of operation, making it suitable for medium-to-high throughput screening. The typical dose-response curve shown in the figure (with a clear EC 50 and wide window) demonstrates that the model can be used for:
①Agonist activity determination
②Antagonist competitive inhibition evaluation
③Relative potency comparison of ligands (e.g., C3a vs. TLQP-21)
2.CHO-K1 Human C3aR β-Arrestin Cell Line(RQP71386)
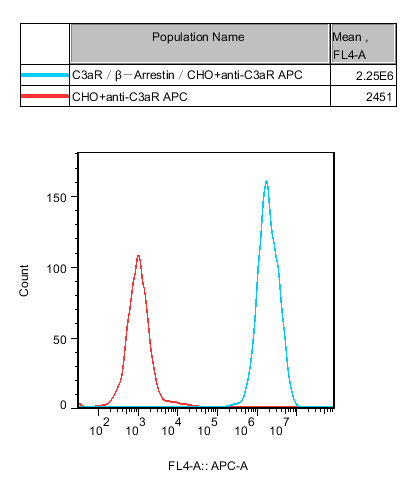
Figure 3. Validation that the cell line stably co-expresses the C3aR and β-Arrestin fusion reporter system, ensuring specificity in functional coupling.
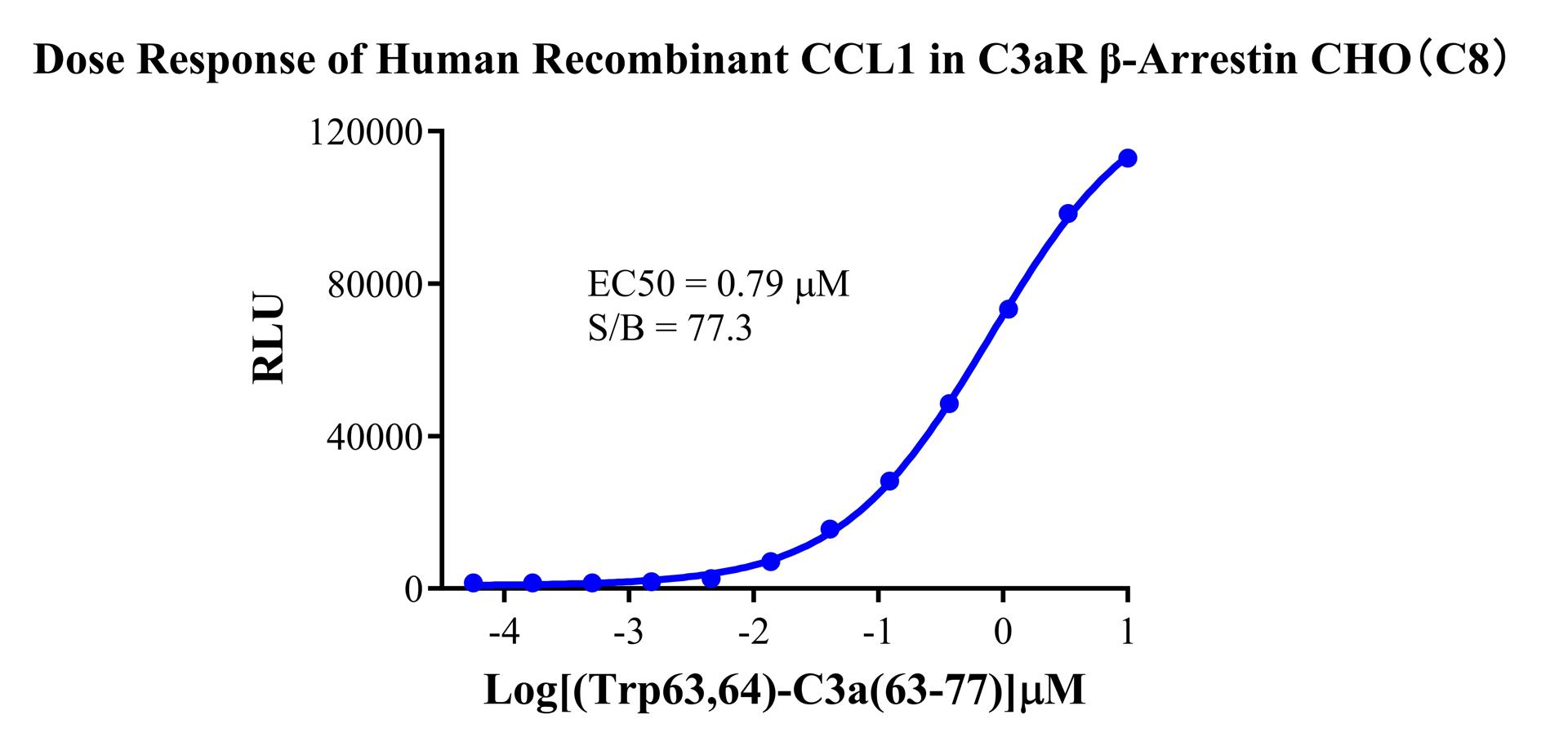
Figure 4. Dose-response experiment conducted using the synthetic peptide [Trp⁶³,⁶⁴]-C3a (63-77) (a stable, highly active C3aR agonist). This model reflects receptor activation by detecting β-Arrestin recruitment and offers the following advantages:
①Homogeneous Assay: No wash steps required, suitable for High-Throughput Screening (HTS).
②Capturing Biased Signaling: Capable of distinguishing between G-protein-dependent and β-Arrestin-dependent agonists.
③Wide Dynamic Range: The sigmoidal curve shown in Figure 6 features a steep slope and large window, making it easy to identify compounds with varying potencies.
Summary of Data Advantages:
|
Dimension of Advantage |
Detailed Description |
| Comprehensive Target Coverage | Provides both G-protein pathway (cAMP) and β-Arrestin pathway detection models for a full-spectrum evaluation of C3aR activity. |
| Mature Detection Platforms | Both HTRF cAMP and β-Arrestin recruitment are "gold standard" methods for GPCR drug screening, ensuring reliable and publishable data. |
| Superior Model Performance | Validation data demonstrates high expression levels, excellent dose-dependency, and wide detection windows, ensuring accuracy and reproducibility of screening results. |
| Flexible Ligand Options | Compatible with the natural ligand C3a, the long-acting ligand TLQP-21, or the synthetic agonist [Trp⁶³,⁶⁴]-C3a (63-77) to suit various experimental objectives. |
| Ready-to-Use Products | Cell models undergo rigorous quality control and functional validation, allowing for immediate experimental use upon arrival to shorten R&D cycles. |
Ⅴ.Conclusion
As a critical hub linking the complement system and immune regulation, C3aR is expanding its reach from traditional inflammatory targets into emerging fields such as cancer immunotherapy. Whether developing small molecule antagonists and monoclonal antibodies or investigating biased signaling mechanisms, stable and reliable functional cell models are the cornerstone of success. Leveraging its profound expertise in the field of GPCR cell models, ReqBio provides two high-performance tool cell lines for C3aR target research, empowering the discovery and clinical translation of innovative drugs worldwide.
news recommendation
CD3E: The T-Cell Activation Switch and Cell Models for Targeted Therapeutics
NFAT Signaling Pathway: A Critical Target for Drug R&D and Cell Models
TSHR Cell Model for Thyroid Cancer Drug Screening | GPCR Assay Platform-Reqbio
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore



