
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Professional technology integration to support the entire R&D process
Home>
Resources>
Learning>
Articles>
From Inflammation to Cancer: Biological Functions of the CCL2-CCR2 Axis and New Progress in Targeted Drug R&D
From Inflammation to Cancer: Biological Functions of the CCL2-CCR2 Axis and New Progress in Targeted Drug R&D
In the frontier exploration of tumor immunotherapy, beyond classical immune checkpoints, the cell migration network mediated by chemokines and their receptors is gradually becoming a new focus for drug R&D. Among them, the CCL2-CCR2 axis, as the core pathway regulating monocyte recruitment, plays a vital role in tumorigenesis, progression, and metastasis. Drugs targeting this axis hold the promise of remodeling the tumor microenvironment and reversing immunosuppression. Leveraging a mature GPCR drug screening platform, Reqbio has developed a series of cell models covering the CCR2 target, aiming to provide high-efficiency and reliable early-stage R&D tools for researchers worldwide.
I. The CCL2-CCR2 Axis: A Key Bridge Connecting Inflammation and Cancer
CCL2, also known as Monocyte Chemoattractant Protein-1 (MCP-1), is one of the earliest discovered and most extensively studied members of the CC chemokine family. It is primarily secreted by immune cells, tumor cells, and tumor-associated stromal cells (such as fibroblasts and endothelial cells). Various pro-inflammatory cytokines, including IL-1, TNF-α, and IFN-γ, can induce the expression of CCL2, making it a pivotal molecule bridging inflammation and cancer.
The primary receptor for CCL2 is CCR2, a member of the G protein-coupled receptor (GPCR) family. CCR2 exists in two isoforms—CCR2A and CCR2B—which may function through distinct downstream signaling pathways. Upon CCL2 binding to CCR2, the receptor undergoes dimerization and internalization, activating multiple signaling cascades including JAK/STAT, PI3K/MAPKs, and PI3K/Akt/NF-κB. These pathways ultimately regulate processes such as cytokine production, cell proliferation, survival, migration, and angiogenesis.
II. The Dual Role of the CCL2-CCR2 Axis in Cancer Immunity: A Behind-the-Scenes Driver of Tumor Progression
In the tumor microenvironment (TME), the CCL2-CCR2 axis promotes tumor progression primarily through the following mechanisms:
1. Recruiting Immunosuppressive Cells to Build a "Protective Shield"
CCL2 is one of the most potent chemoattractants for monocytes and macrophages. By recruiting CCR2+ monocytes into the tumor microenvironment and promoting their differentiation into Tumor-Associated Macrophages (TAMs) and Myeloid-Derived Suppressor Cells (MDSCs), the CCL2-CCR2 axis directly constructs an immunosuppressive network. These cells secrete factors such as IL-10 and TGF-β, which inhibit the killing function of CD8+ T cells, helping the tumor evade immune surveillance.
2. Promoting Angiogenesis to "Fuel" the Tumor
TAMs secrete Vascular Endothelial Growth Factor (VEGF), which interacts with CCR2+ vascular endothelial cells to promote the formation of new blood vessels within the tumor, providing oxygen and nutrients for rapid tumor growth.
3. Driving Tumor Metastasis to Establish "Colonies"
The CCL2-CCR2 axis functions at multiple steps of the metastatic cascade:
Local Invasion: Stimulating cancer cells to invade surrounding tissues.
Survival in Circulation: Helping tumor cells spread to distant organs along chemotactic gradients.
Formation of Pre-metastatic Niches: In distant organs (such as the lungs and liver), CCL2 recruits monocytes that differentiate into Metastasis-Associated Macrophages (MAMs), creating a microenvironment suitable for the colonization and growth of circulating tumor cells.
Due to its core driving role in tumor progression and metastasis, the CCL2-CCR2 axis has become an attractive intervention target in cancer therapy.
Ⅲ.Global Progress of Drugs Targeting CCL2/CCR2
Currently, a variety of drugs targeting CCL2 or CCR2 (including small molecule inhibitors and neutralizing antibodies) have entered the clinical research stage. Their indications cover oncology, inflammatory diseases, and autoimmune disorders.
|
Drug Name |
Developer |
Phase |
Drug Type |
Target |
Indication |
|
CCX-140 |
ChemoCentryx |
Phase II |
Small Molecule | CCR2 | Diabetic Nephropathy |
| CNTX-6970 | C.H. Boehringer Sohn |
Phase II |
Small Molecule | CCR2 | Knee Osteoarthritis |
| STX-0712 | Solu Therapeutics |
Phase I |
ADC | CCR2 | Tumors, Hematological Diseases |
| RS-102895 | Roche Bioscience |
Preclinical |
Small Molecule | CCR2 | Atherosclerosis, Asthma |
| 2,5-Dihydroxybenzoic Acid | China Medical University |
Preclinical |
Small Molecule | CCL2 & CCR2 | Fatty Liver Disease |
| ST-MT@lipo2 | Fujian Medical University Union Hospital |
Preclinical |
Liposome | CCL2 & CCR2 | Myocardial Ischemia |
From the perspective of R&D trends, in addition to traditional small molecule inhibitors, Antibody-Drug Conjugates (ADCs) and novel delivery systems are emerging as new directions for targeting the CCL2-CCR2 axis.
Ⅳ.Accelerating CCR2 Drug R&D: Reqbio’s One-Stop Cell Model Solutions
In GPCR drug R&D, reliable functional screening models are key to discovering lead compounds and optimizing drug candidates. Leveraging years of experience, Reqbio has successfully developed and validated a series of cell models targeting the CCR2 receptor, meeting the diverse needs of different screening stages.
Reqbio CCR2 Cell Model Product List:
|
Cell Model Name |
Catalog No. |
Core Application |
Features |
| CHO-K1 Human CCR2 β-Arrestin Cell Line | RQP71394 | Agonist/Antagonist Screening | Based on β-Arrestin recruitment, suitable for HTS |
| CHO-K1 Human CCR2 Cell Line | RQP71350 | cAMP Signaling Detection | Suitable for assessing G protein signaling activity |
| CHO-K1 Human CCR2 Gα15 Cell Line | RQP71335 | Calcium Flux Detection | Suitable for detecting GPCR-mediated calcium release |
| HEK293 Human CCR2 Gα16 Cell Line | RQP71055 | Multi-Functional Signaling Detection | Suitable for mechanism studies of different downstream pathways |
Product Validation Data Display:
1.Functional Validation of CHO-K1 Human CCR2 β-Arrestin Cell Line(RQP71394)
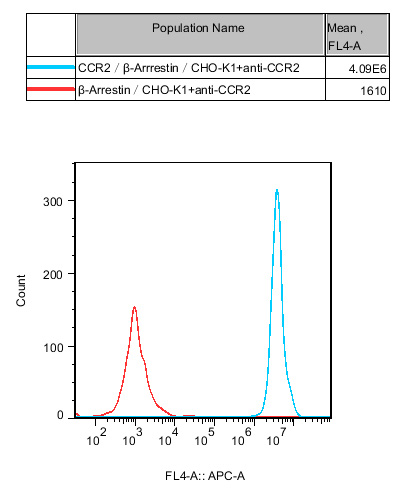
Figure 1. Recombinant CCR2/β-Arrestin/CHO constitutively expressing CCR2.
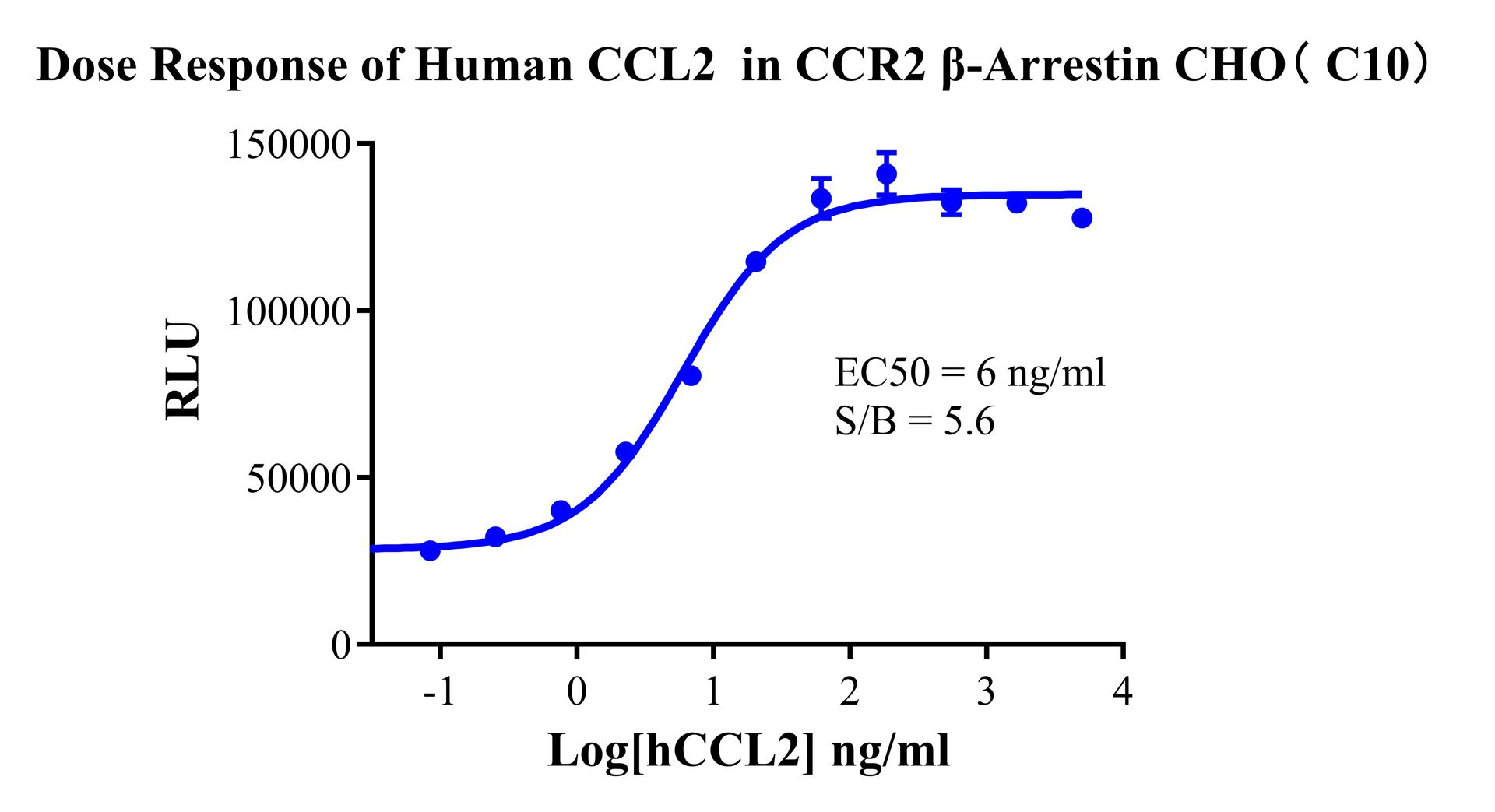
Figure 2. Dose Response of Human CCL2 in CCR2 β-Arrestin CHO(C10).
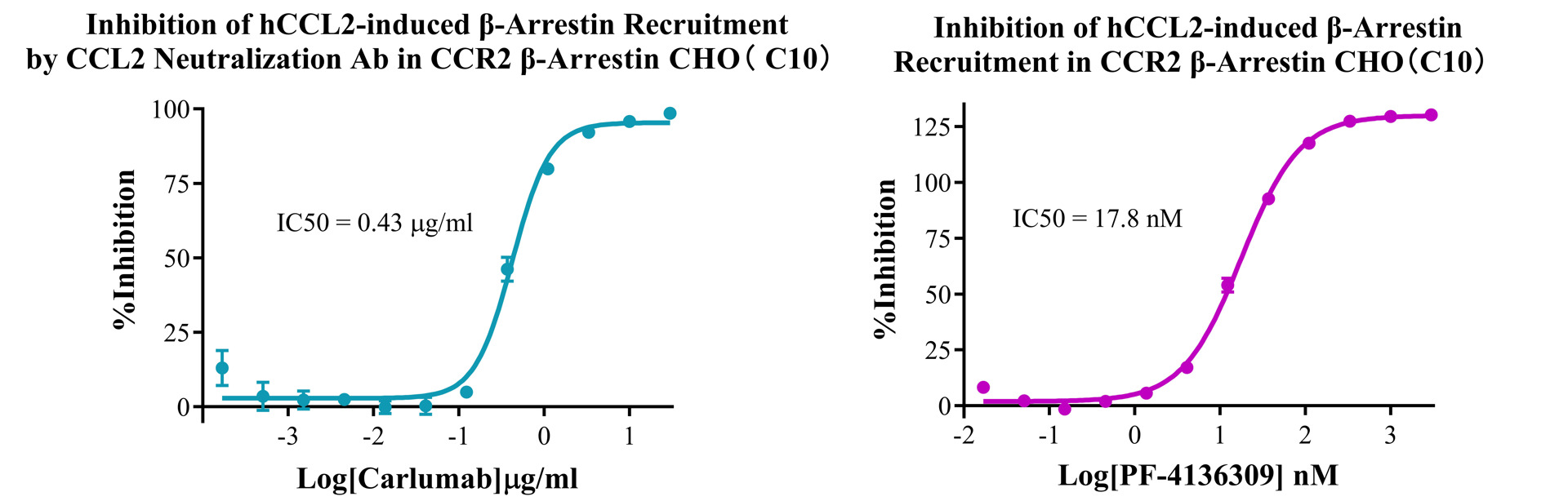
Figure 3.Inhibition of hCCL2-induced β-Arrestin Recruitment by CCL2 Neutralization Ab in CCR2 β-Arrestin CHO(C10). Inhibition of hCCL2-induced β-Arrestin Recruitment in CCR2 β-Arrestin CHO(C10).
2.Functional Validation of CHO-K1 Human CCR2 Cell Line(RQP71350)
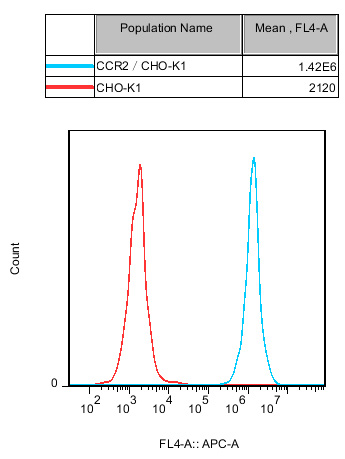
Figure 4. Recombinant CCR2/CHO stably expressing CCR2.
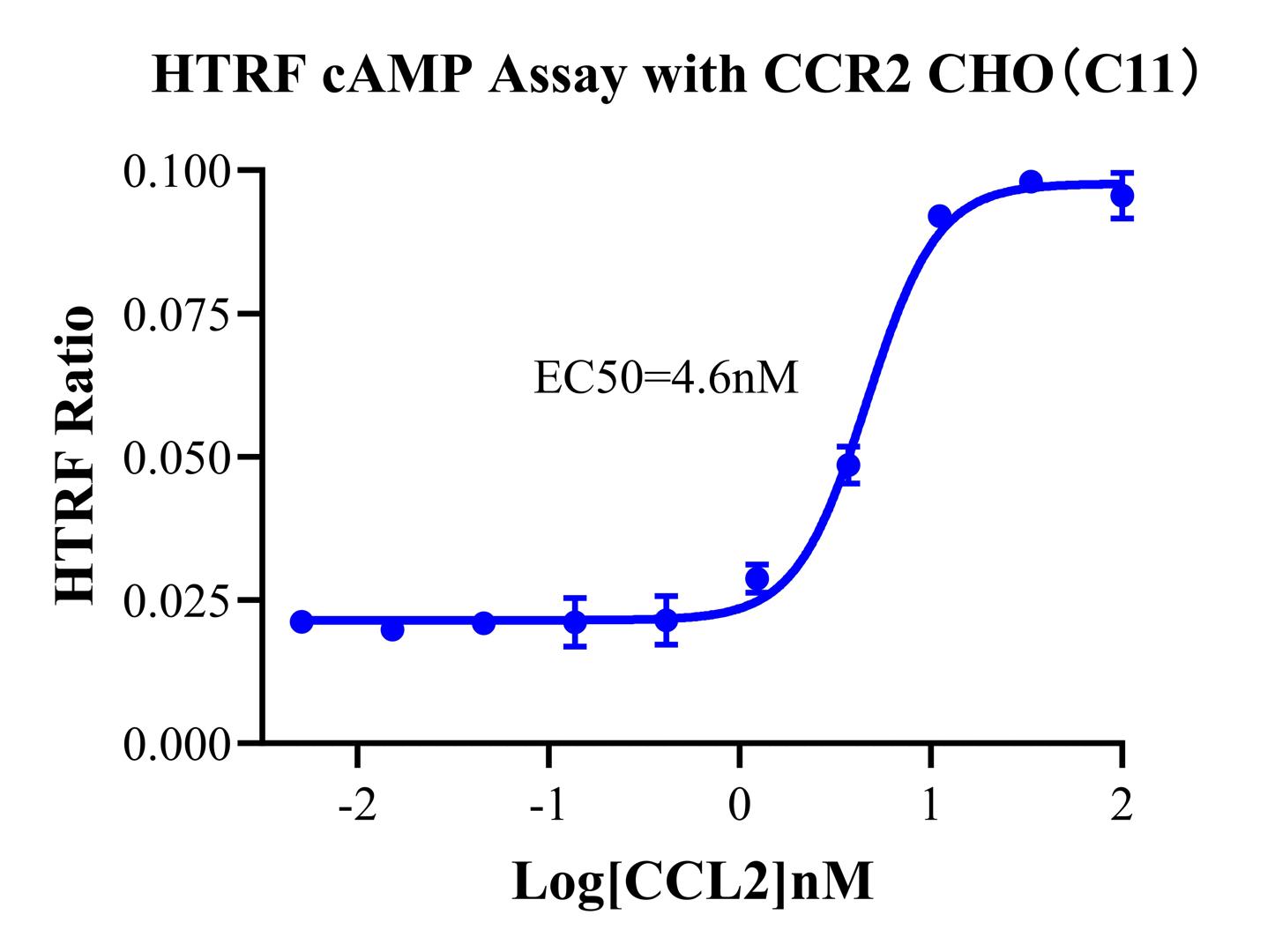
Figure 5. HTRF cAMP Assay with CCR2 CHO(C11).
Product Core Advantages:
High Sensitivity and Stability: Rigorous functional validation ensures excellent consistency and reproducibility of experimental data.
Diverse Detection Platforms: Support for multiple assay modes, including $\beta$-arrestin recruitment, cAMP inhibition, and calcium flux, meeting diverse screening requirements.
Ready-to-Use Products: Streamlined experimental workflows shorten R&D cycles, helping customers accelerate early-stage drug discovery.
Ⅴ.Conclusion
As our understanding of the tumor microenvironment deepens, targeting the CCL2-CCR2 axis is transitioning from concept to clinical reality. Whether you are developing small molecule inhibitors, neutralizing antibodies, or novel ADCs, reliable screening models are the cornerstone of success. Reqbio remains committed to focusing on frontier targets in GPCRs and immuno-oncology, providing high-quality biological reagents and cell models to empower global innovative drug discovery.
news recommendation
NFAT Signaling Pathway: A Critical Target for Drug R&D and Cell Models
TSHR Cell Model for Thyroid Cancer Drug Screening | GPCR Assay Platform-Reqbio
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore



