
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
Jurkat E6.1 Human NKG2A Effector Reporter Cell
Cat. No: RQP74241
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74241 |
| Product Name | Jurkat E6.1 Human NKG2A Effector Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | suspension |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | RPMI-1640+10%FBS+1μg/ml puromycin+10 μg/ml Blasticidin+ 800 μg/ml Hygromycin B |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
NKG2A is a member of the C-type lectin superfamily, specifically a single-pass type II transmembrane glycoprotein comprising an intracellular domain, a transmembrane domain, and an extracellular lectin-like domain. The intracellular domain contains two ITIMs (immunoreceptor tyrosine-based inhibitory motifs) that participate in inhibitory signal transduction. NKG2A serves as a critical immune checkpoint for natural killer (NK) cells and CD8+ T cells. NKG2A is expressed as a heterodimer with CD94—which also belongs to the C-type lectin superfamily—and the ligand for this NKG2A/CD94 heterodimeric receptor is a non-classical MHC Class I molecule. Upon binding, the NKG2A/CD94 complex transmits inhibitory signals to NK cells and CD8+ T cells; consequently, blocking NKG2A effectively unleashes the cytotoxic functions of these lymphocytes.
Binding of the NKG2A/CD94 receptor to the peptide-presenting molecule HLA-E triggers the phosphorylation of the ITIMs within NKG2A. These phosphorylated ITIMs are responsible for recruiting and activating the intracellular phosphatases SHP-1 and SHP-2, thereby suppressing the activating signals generated by receptors such as the T-cell receptor (TCR) and NKG2D. In contrast to classical HLA molecules—which are often lost—the expression of HLA-E is frequently upregulated in tumor cells. The interaction between NKG2A and HLA-E contributes to tumor immune evasion, and NKG2A-mediated mechanisms are currently being leveraged to develop potential anti-tumor therapeutic strategies. Furthermore, disrupting the interaction between NKG2A and its ligand has been demonstrated to effectively enhance anti-tumor immune responses. The expression level of NKG2A correlates with disease severity in patients with COVID-19. Additionally, NKG2A is implicated in the pathological processes of various other immune-mediated conditions, including autoimmune diseases, inflammatory disorders, parasitic infections, and transplant rejection. These findings suggest that NKG2A represents a novel therapeutic target for a wide spectrum of immune-mediated diseases. Furthermore, increasing evidence indicates that NKG2A also plays a significant role in other immune-related diseases, including viral infections, autoimmune diseases, inflammatory diseases, parasitic infections, and transplant rejection.
The Jurkat E6.1 Human NKG2A Effector Reporter Cell Model—effectively simulates the signal transduction process of NKG2A *in vivo*. The underlying principle is illustrated in the figure below.
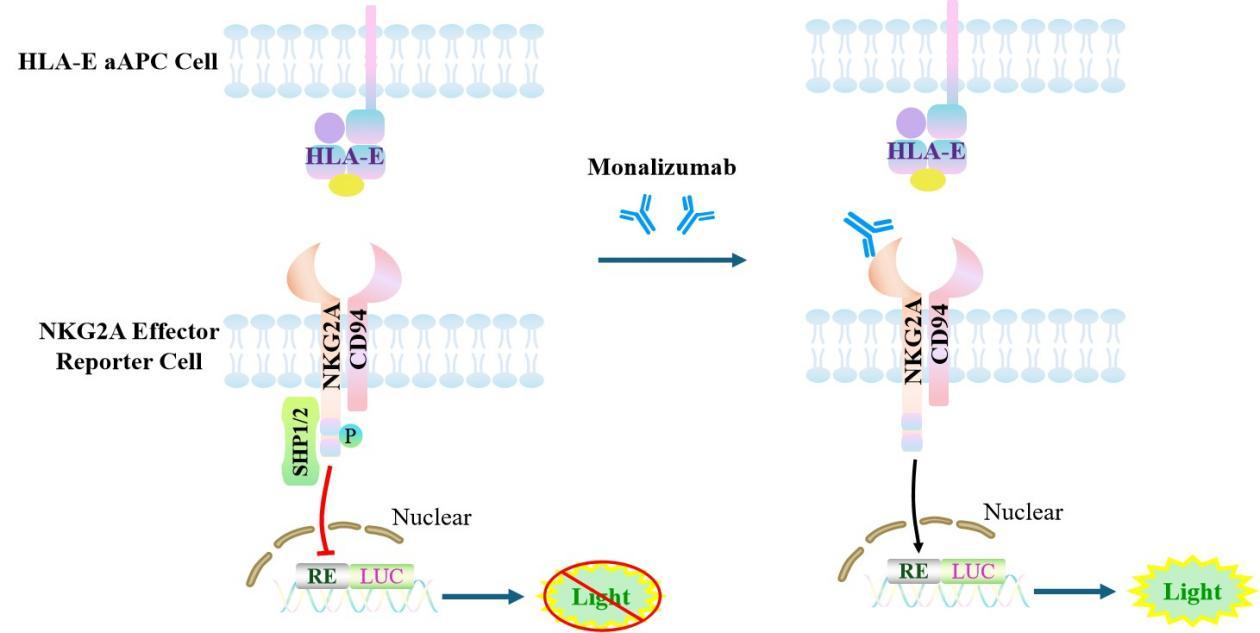
Figure 1. Schematic Diagram of the Jurkat E6.1 Human NKG2A Effector Reporter Cell Model
| Classification | Co-Inhibitory |
| Family | C‑type lectin‑like receptor (CTLR) family |
| Gene Name | KLRC1 |
| Gene Aliases | NKG2;NKG2-A;NKG2-B;CD159a |
| Gene ID | 3821 |
| Accession Number | NM_002259.5 |
| UniProt Number | P26715 |
| Protein Name | NKG2-A/NKG2-B type II integral membrane protein |
| Protein Aliases | CD159 antigen-like family member A;NK cell receptor A;NKG2-A/B-activating NK receptor |
| Target Species | Human |
| Host cell | Jurkat E6.1 |
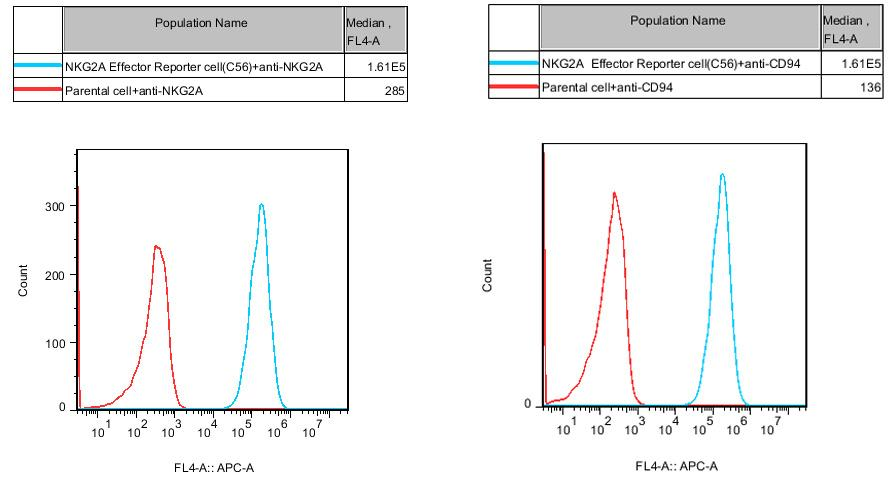
Figure 2. Recombinant NKG2A Effector Reporter Cell expressing NKG2A and CD94.

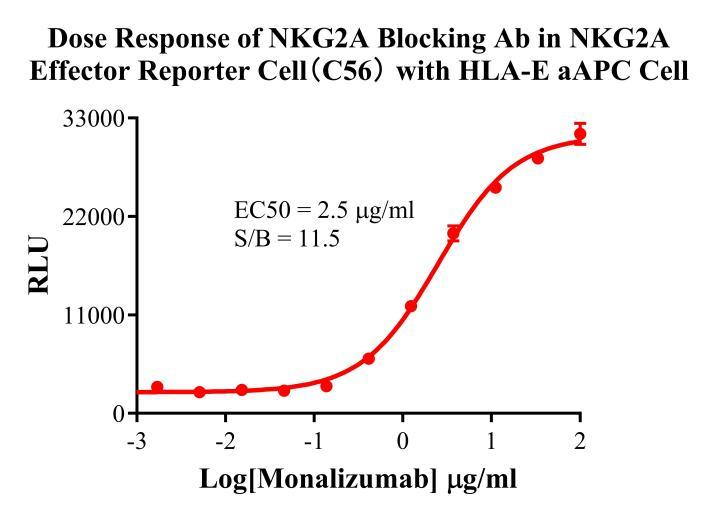
Figure 3. Dose Response of NKG2A Blocking Ab in NKG2A Effector Reporter Cell(C56) With HLA-E aAPC Cell.
Cell Passage Procedures
1.This cell line grows in suspension.
2.Upon receipt, cells should be thawed immediately or stored in liquid nitrogen until use.
3.Before thawing, pre-warm the water bath and culture medium to 37 °C, and prepare a small amount of dry ice.
4.Remove the cryovial from storage and transport it to the cell culture laboratory on dry ice.
5.Rapidly thaw the cells in a 37 °C water bath. Once the cells are completely thawed, spray the cryovial with 70% ethanol for disinfection and transfer it to a biosafety cabinet.
6.Add 10 mL of pre-warmed culture medium into a 15 mL centrifuge tube. Transfer the contents of the cryovial into the tube and centrifuge at 1000 rpm for 5 minutes.
7.Carefully discard the supernatant. Resuspend the cell pellet in 5 mL of pre-warmed culture medium by gentle pipetting. Immediately perform cell counting and adjust the cell density to 3–6 × 10⁵ cells/mL based on the counting results, then transfer the cells into a culture flask.
8.Count the cells every 1–2 days. When the cell density exceeds 1 × 10⁶ cells/mL, passage the cells promptly or add fresh culture medium. Maintain the cell density between 2 × 10⁵ and 1 × 10⁶ cells/mL.
Suspension Cell Cryopreservation Procedure:
1.Collect 8 × 10⁶ cells, centrifuge, and discard the supernatant.
2.Add 1 mL of cell freezing medium (90% FBS + 10% DMSO) and gently pipette to mix thoroughly. Transfer the suspension into a cryovial.
3.Immediately place the cryovial into a controlled-rate freezing container (Nalgene 5100-0001), fill with isopropanol up to the indicated level, and store at −80 °C.
4.After 24 hours, transfer the cryovial to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore