
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
THP-1 Human ISRE-Luc/NFκB-SEAP Cell
Cat. No: RQP74097
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Assay Data
Cell Culture
| Cat. No | RQP74097 |
| Product Name | THP-1 Human ISRE-Luc/NFκB-SEAP Cell |
| Product Type | Reporter Cell |
| Culture Properties | suspension |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | RPMI-1640+ 10% heat-inactivated fetal bovine serum+100 μg/ml Normocin+10 µg/ml blasticidin+100 ug/ml Zeocin |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
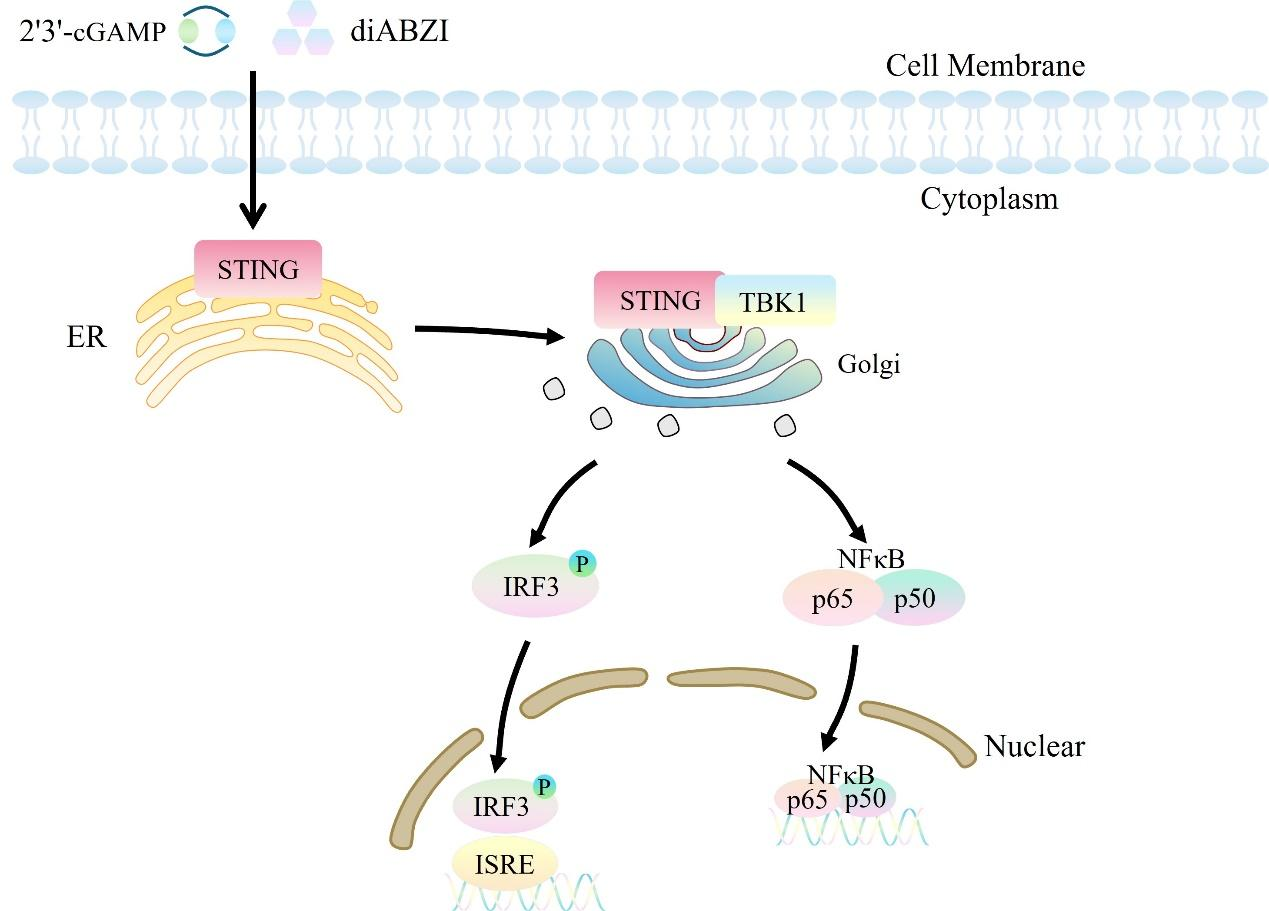
STING (stimulator of interferon genes), also known as MPYS, TMEM173, MITA, and ERIS, is a transmembrane protein that serves as a direct sensor of cyclic dinucleotides (CDNs). CDNs are second messenger molecules commonly found in bacterial signal transduction and act as triggers for defense mechanisms in mammalian cells. When bacterial pathogens attack, CDNs released into the cell directly bind to STING. Activated STING interacts with Tank-binding kinase-I (TBK1) to induce the activation of interferon regulatory factor 3 (IRF3) dimers. These dimers then bind to interferon-stimulated response elements (ISRE) in the cell nucleus, leading to the production of IFN-α/β. The production of IFN-β is a hallmark of STING activation. Signaling from the innate immune system within the tumor microenvironment (TME) is a key factor in driving the activation of tumor-specific T cells and the infiltration of tumor-infiltrating lymphocytes (TILs). Among these, type I IFN is particularly critical for activating tumor-specific T cells. Therefore, STING agonists can activate immune-stimulating cells such as dendritic cells by inducing the expression of type I interferon genes, alter the tumor microenvironment, and induce the generation of tumor-specific T cells, thereby achieving anti-tumor effects through modulation of the immune system.
ISRE-Luc NFκB-SEAP THP-1 reporter gene cells are designed for drug discovery targeting STING agonists and antagonists. Leveraging the principle that the STING protein regulates interferon (IFN) and the expression of its downstream genes, two inducible reporter gene systems have been constructed in THP-1 cells: one is the interferon-stimulated response element (ISRE), which is linked to a secreted luciferase (Luc). This system generates chemiluminescent signals upon substrate stimulation and can be used to evaluate the IFN pathway, such as type I IFNs, RLR (RIG-I-like receptor), CDS (cytosolic dsDNA sensor), or STING agonists. The other system targets the NFκB pathway, with a secreted embryonic alkaline phosphatase (SEAP) reporter gene downstream, which responds to stimuli related to the NFκB pathway. The ISRE-Luc NFκB-SEAP THP-1 reporter gene drug target model effectively mimics the signal transduction process.

Figure 1. Schematic diagram of the THP-1 Human ISRE-Luc/NFκB-SEAP Cell model.
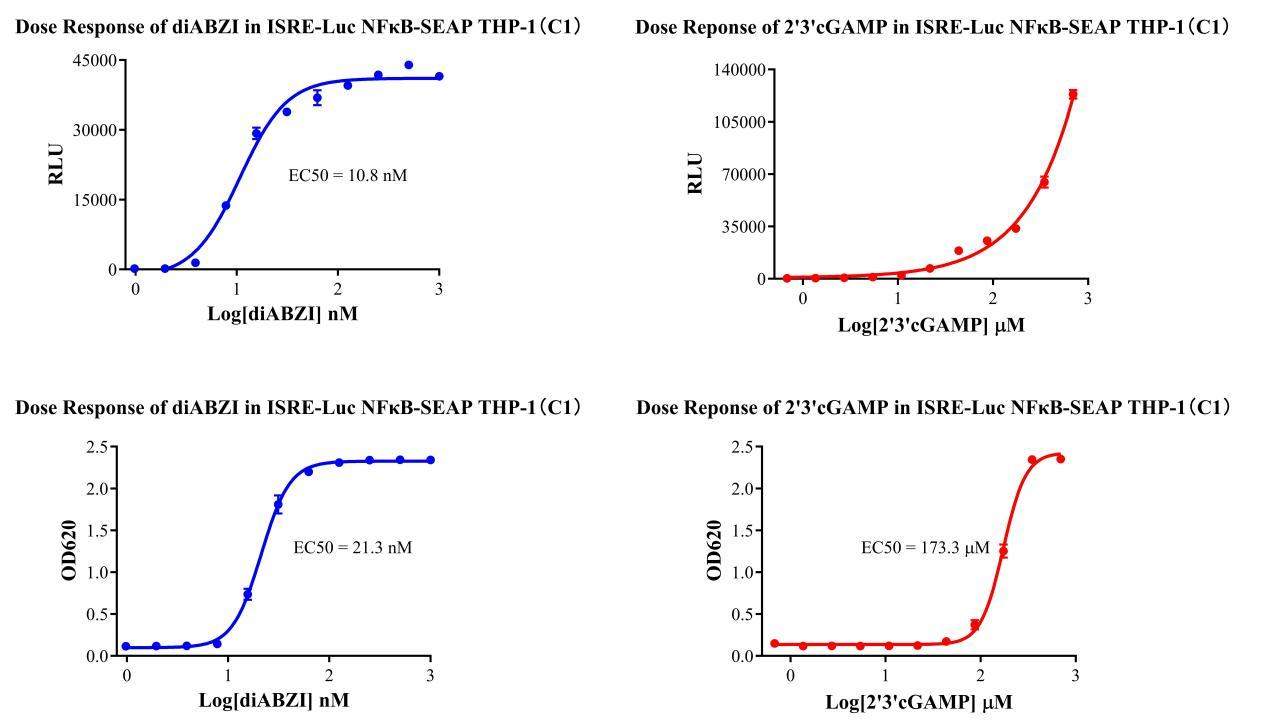
Figure 2. Verification Results of the STING Stimulation Assay.
Cell Passage Procedures
1.This cell line grows in suspension.
2.Upon receipt, cells should be thawed immediately or stored in liquid nitrogen until use.
3.Before thawing, pre-warm the water bath and culture medium to 37 °C, and prepare a small amount of dry ice.
4.Remove the cryovial from storage and transport it to the cell culture laboratory on dry ice.
5.Rapidly thaw the cells in a 37 °C water bath. Once the cells are completely thawed, spray the cryovial with 70% ethanol for disinfection and transfer it to a biosafety cabinet.
6.Add 10 mL of pre-warmed culture medium into a 15 mL centrifuge tube. Transfer the contents of the cryovial into the tube and centrifuge at 1000 rpm for 5 minutes.
7.Carefully discard the supernatant. Resuspend the cell pellet in 5 mL of pre-warmed culture medium by gentle pipetting. Immediately perform cell counting and adjust the cell density to 3–6 × 10⁵ cells/mL based on the counting results, then transfer the cells into a culture flask.
8.Count the cells every 1–2 days. When the cell density exceeds 1 × 10⁶ cells/mL, passage the cells promptly or add fresh culture medium. Maintain the cell density between 2 × 10⁵ and 1 × 10⁶ cells/mL.
Suspension Cell Cryopreservation Procedure:
1.Collect 8 × 10⁶ cells, centrifuge, and discard the supernatant.
2.Add 1 mL of cell freezing medium (90% FBS + 10% DMSO) and gently pipette to mix thoroughly. Transfer the suspension into a cryovial.
3.Immediately place the cryovial into a controlled-rate freezing container (Nalgene 5100-0001), fill with isopropanol up to the indicated level, and store at −80 °C.
4.After 24 hours, transfer the cryovial to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore