
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
Jurkat E6.1 Human TREM1 Effector Reporter Cell
Cat. No: RQP74204
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74204 |
| Product Name | Jurkat E6.1 Human TREM1 Effector Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | suspension |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | RPMI-1640+10%FBS+1μg/ml puromycin+800μg/ml Hygromycin B |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
TREM1 is a member of the immunoglobulin superfamily—a group of cell-surface receptors characterized by related extracellular Ig-like domains. In addition to its extracellular Ig domain, it comprises a transmembrane domain containing a conserved lysine residue and a short cytoplasmic domain devoid of signaling motifs. Studies suggest that High Mobility Group Box 1 (HMGB1), Peptidoglycan Recognition Protein 1 (PGLYRP1), Heat Shock Protein 70 (HSP70), CD177, Actin, and extracellular Cold-Inducible RNA-Binding Protein (eCIRP) may serve as natural ligands for TREM-1; notably, all of these endogenous ligands are associated with inflammatory responses.
TREM1 signal transduction relies on its association with the immunoreceptor adaptor protein DNAX-activating protein of 12 kDa (DAP12). TREM-1 possesses a positively charged lysine residue within its transmembrane domain, which pairs with a negatively charged aspartate residue on DAP12. Upon receptor activation, the Immunoreceptor Tyrosine-based Activation Motif (ITAM) within DAP12 undergoes phosphorylation, thereby recruiting and activating Spleen Tyrosine Kinase (Syk). Syk subsequently activates a multitude of downstream signaling pathways—including the PI3K/AKT, Ras/ERK/MAPK, and NF-κB pathways—and triggers the phosphorylation of Phospholipase C, ultimately leading to increased intracellular calcium levels and the secretion of pro-inflammatory cytokines.
The TREM1 Effector Reporter model effectively recapitulates the in vivo signal transduction processes of TREM1; the underlying principle is illustrated in the figure below.
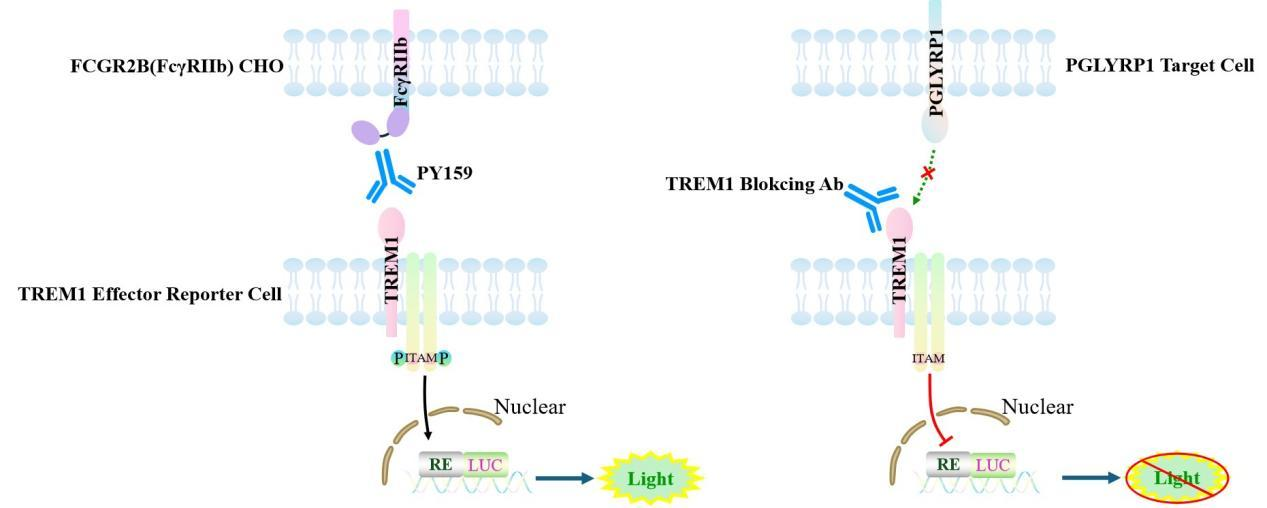
Figure 1. Schematic Diagram of the Jurkat E6.1 Human TREM1 Effector Reporter Cell Model
| Classification | Co-Stimulatory |
| Family | TREM family |
| Gene Name | TREM1 |
| Gene Aliases | TREM-1;CD354 |
| Gene ID | 54210 |
| Accession Number | NM_018643.5 |
| UniProt Number | Q9NP99 |
| Protein Name | TREM-1 |
| Protein Aliases | Triggering receptor expressed on monocytes 1 |
| Target Species | Human |
| Host cell | Jurkat E6.1 |
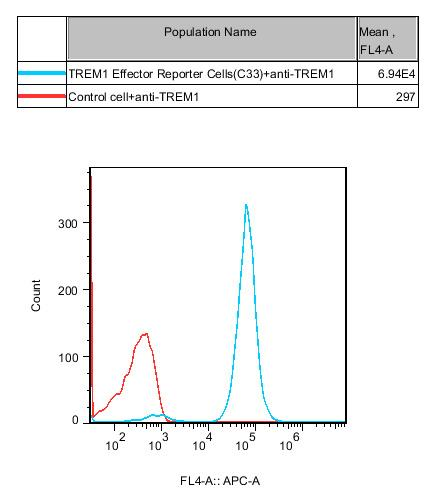
Figure 2. Recombinant TREM1 Effector Reporter Cell constitutively expressing TREM1.

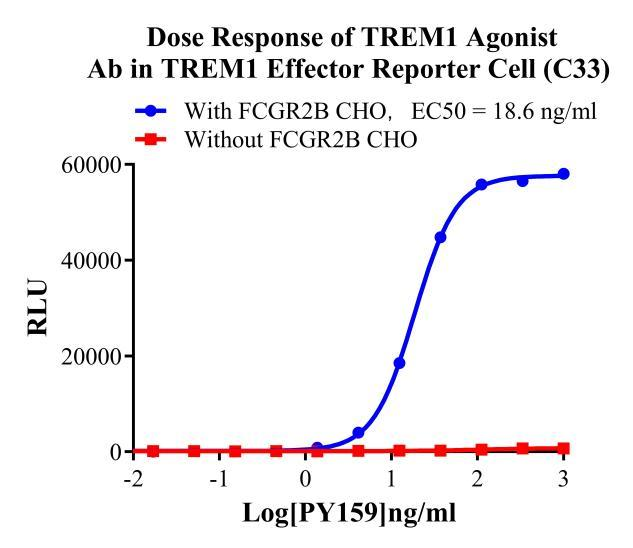
Figure 3. Dose Response of TREM1 Agonist Ab in TREM1 Effector Reporter Cell(C33).
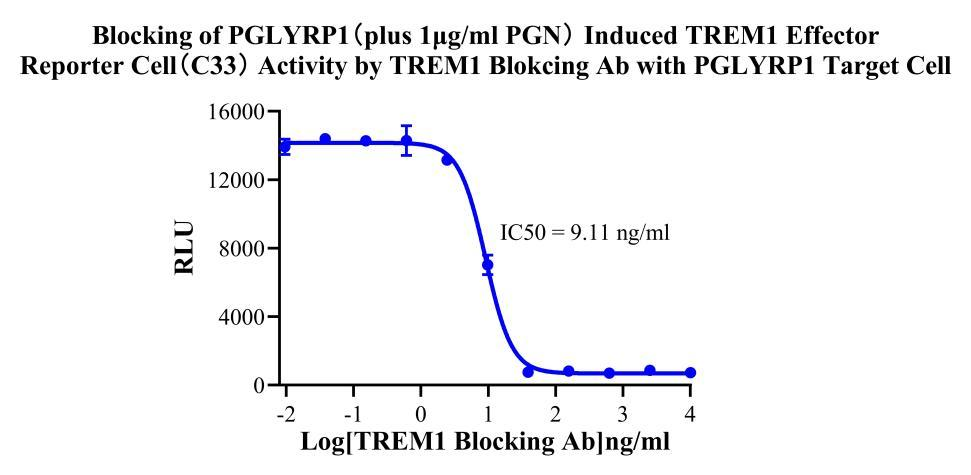
Figure 4. Blocking of PGLYRP1 (plus 1μg/ml PGN)induced TREM1 Effector Reporter Cell(C33) Activity by TREM1 Blokcing Ab with PGLYRP1 Target Cell .
Cell Passage Procedures
1.This cell line grows in suspension.
2.Upon receipt, cells should be thawed immediately or stored in liquid nitrogen until use.
3.Before thawing, pre-warm the water bath and culture medium to 37 °C, and prepare a small amount of dry ice.
4.Remove the cryovial from storage and transport it to the cell culture laboratory on dry ice.
5.Rapidly thaw the cells in a 37 °C water bath. Once the cells are completely thawed, spray the cryovial with 70% ethanol for disinfection and transfer it to a biosafety cabinet.
6.Add 10 mL of pre-warmed culture medium into a 15 mL centrifuge tube. Transfer the contents of the cryovial into the tube and centrifuge at 1000 rpm for 5 minutes.
7.Carefully discard the supernatant. Resuspend the cell pellet in 5 mL of pre-warmed culture medium by gentle pipetting. Immediately perform cell counting and adjust the cell density to 3–6 × 10⁵ cells/mL based on the counting results, then transfer the cells into a culture flask.
8.Count the cells every 1–2 days. When the cell density exceeds 1 × 10⁶ cells/mL, passage the cells promptly or add fresh culture medium. Maintain the cell density between 2 × 10⁵ and 1 × 10⁶ cells/mL.
Suspension Cell Cryopreservation Procedure:
1.Collect 8 × 10⁶ cells, centrifuge, and discard the supernatant.
2.Add 1 mL of cell freezing medium (90% FBS + 10% DMSO) and gently pipette to mix thoroughly. Transfer the suspension into a cryovial.
3.Immediately place the cryovial into a controlled-rate freezing container (Nalgene 5100-0001), fill with isopropanol up to the indicated level, and store at −80 °C.
4.After 24 hours, transfer the cryovial to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore