
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
Jurkat E6.1 Human GUCY2C Effector Reporter Cell
Cat. No: RQP74545
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74545 |
| Product Name | Jurkat E6.1 Human GUCY2C Effector Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | Suspension |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | DMEM+10%FBS+2 μg/ml Puromycin+ 200 μg/ml Hygromycin B+5 μg/ml Blasticidin |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
Guanylate Cyclase C (GUCY2C/GCC) belongs to the family of receptor guanylyl cyclases (GC)—enzymes capable of converting GTP into the second messenger cGMP. GUCY2C is a type I transmembrane receptor protein highly expressed on the apical membranes of intestinal epithelial cells; it serves as a receptor for the endogenous peptides guanylin and uroguanylin, as well as for the heat-stable enterotoxin produced by *E. coli*. GUCY2C is widely expressed in colorectal cancer cells and other gastrointestinal tumors. Dysregulation—specifically, either a loss or an increase in GUCY2C function—is associated with several conditions characterized by constipation or diarrhea, including Irritable Bowel Syndrome with Constipation (IBS-C), Chronic Idiopathic Constipation (CIC), and Inflammatory Bowel Disease (IBD).
Upon activation by endogenous ligands or the exogenous bacterial toxin STa, GUCY2C catalyzes the production of the second messenger cGMP. This, in turn, regulates ion and fluid secretion in intestinal epithelial cells by activating the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR), thereby maintaining fluid and electrolyte homeostasis. Simultaneously, this signaling pathway contributes to maintaining the integrity of the intestinal epithelial barrier and exerts anti-inflammatory effects, thereby protecting the gut from injury. Furthermore, GUCY2C plays a pivotal role as a tumor suppressor by inhibiting the AKT signaling pathway; it regulates the proliferation and metabolism of intestinal epithelial cells, thereby suppressing the initiation and progression of tumors such as colorectal cancer.
The Jurkat E6.1 Human GUCY2C Effector Reporter Cell model accurately simulates the in vivo GUCY2C signal transduction process.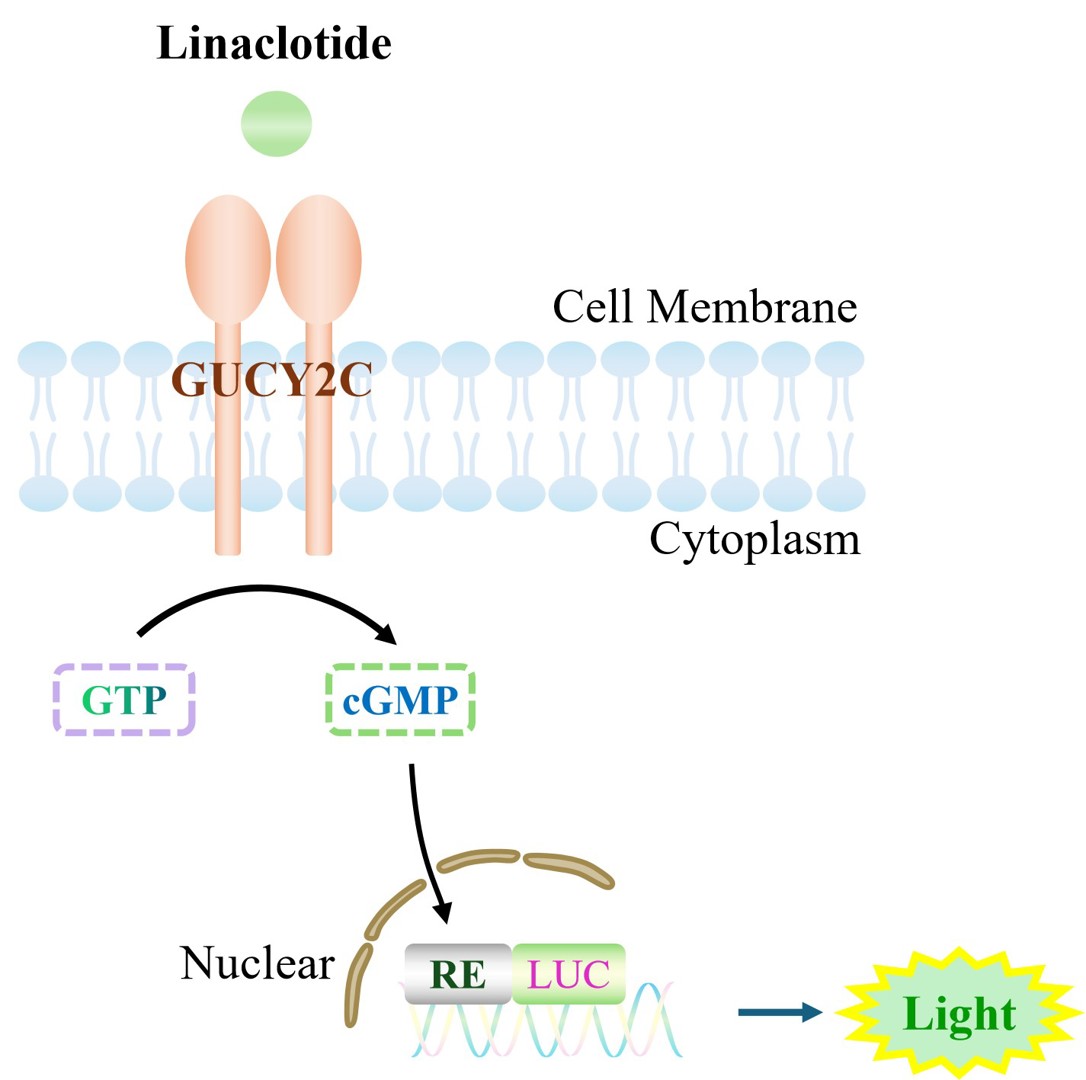
Figure 1. Schematic Diagram of the Jurkat E6.1 Human GUCY2C Effector Reporter Cell Model
| Classification | Cytokine&Growth Factor |
| Family | Receptor guanylyl cyclase family |
| Gene Name | GUCY2C |
| Gene Aliases |
STAR;HSER;GC-C;GCC;
|
| Gene ID | 2984 |
| Accession Number | NM_004963.4 |
| UniProt Number | P25092 |
| Protein Name | Guanylyl cyclase C |
| Protein Aliases | GC-C; STAR; hSTAR; STA receptor; GUC2C; Intestinal guanylate cyclase |
| Target Species | Human |
| Host cell | Jurkat E6.1 |
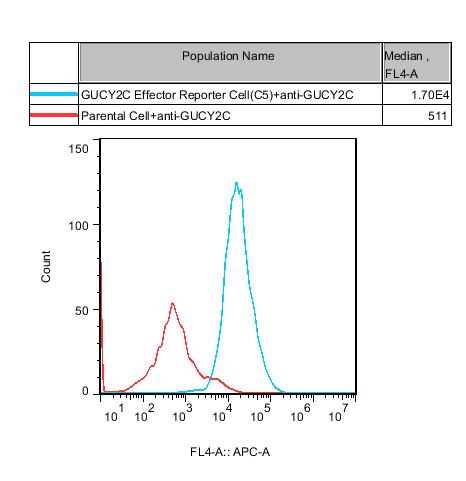
Figure 2. Recombinant GUCY2C Effector Reporter Cell stably expressing GUCY2C.
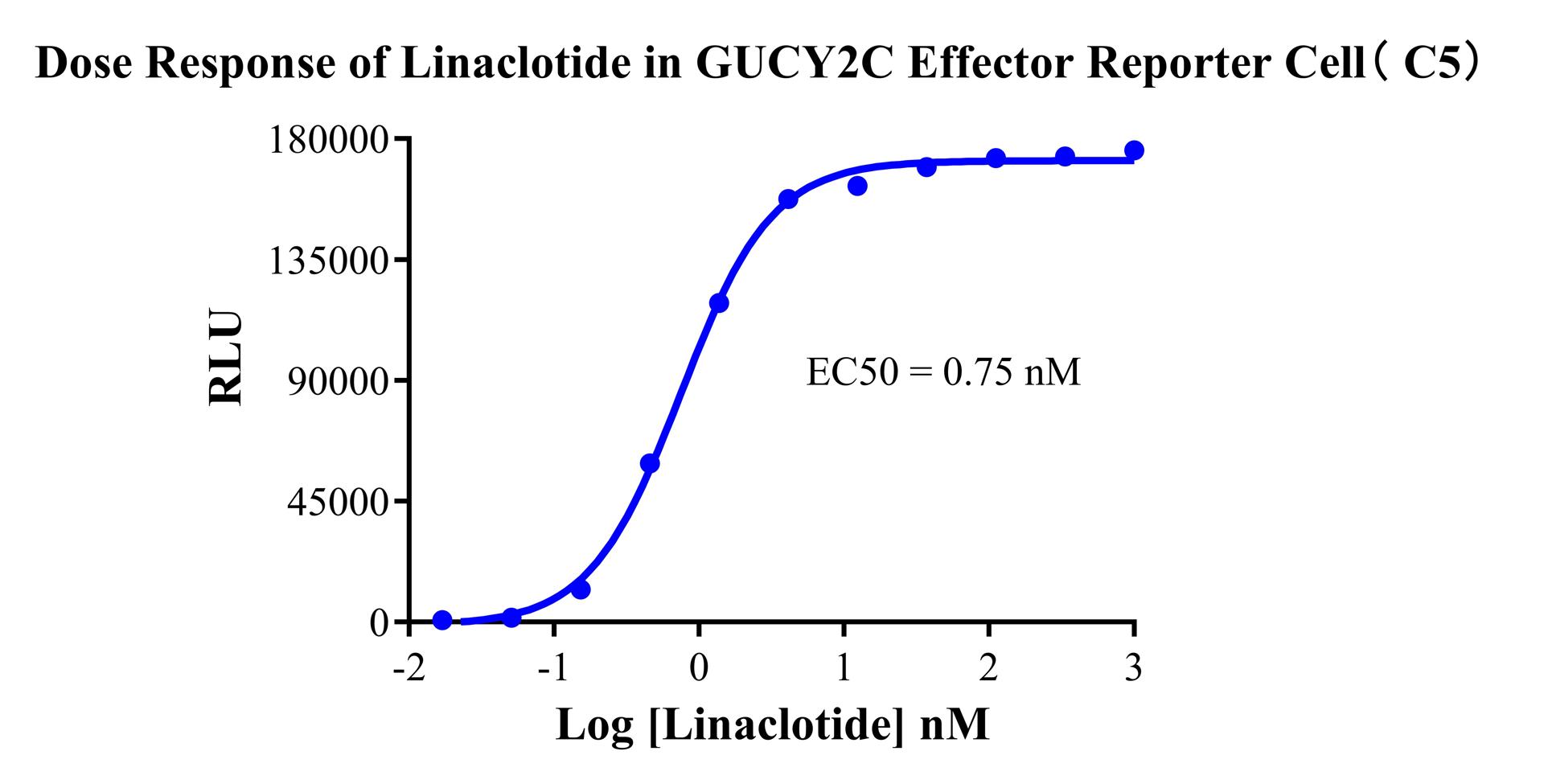
Figure 3. Dose Response of Linaclotide in GUCY2C Effector Reporter Cell (C5).
Cell Passage Procedures
1.This cell line grows in suspension.
2.Upon receipt, cells should be thawed immediately or stored in liquid nitrogen until use.
3.Before thawing, pre-warm the water bath and culture medium to 37 °C, and prepare a small amount of dry ice.
4.Remove the cryovial from storage and transport it to the cell culture laboratory on dry ice.
5.Rapidly thaw the cells in a 37 °C water bath. Once the cells are completely thawed, spray the cryovial with 70% ethanol for disinfection and transfer it to a biosafety cabinet.
6.Add 10 mL of pre-warmed culture medium into a 15 mL centrifuge tube. Transfer the contents of the cryovial into the tube and centrifuge at 1000 rpm for 5 minutes.
7.Carefully discard the supernatant. Resuspend the cell pellet in 5 mL of pre-warmed culture medium by gentle pipetting. Immediately perform cell counting and adjust the cell density to 3–6 × 10⁵ cells/mL based on the counting results, then transfer the cells into a culture flask.
8.Count the cells every 1–2 days. When the cell density exceeds 1 × 10⁶ cells/mL, passage the cells promptly or add fresh culture medium. Maintain the cell density between 2 × 10⁵ and 1 × 10⁶ cells/mL.
Suspension Cell Cryopreservation Procedure:
1.Collect 8 × 10⁶ cells, centrifuge, and discard the supernatant.
2.Add 1 mL of cell freezing medium (90% FBS + 10% DMSO) and gently pipette to mix thoroughly. Transfer the suspension into a cryovial.
3.Immediately place the cryovial into a controlled-rate freezing container (Nalgene 5100-0001), fill with isopropanol up to the indicated level, and store at −80 °C.
4.After 24 hours, transfer the cryovial to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore