
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
Jurkat E6.1 Human ADCP Bioassay Effector Cell FcγRIIa (R variant) -NFAT
Cat. No: RQP74004
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74004 |
| Product Name | Jurkat E6.1 Human ADCP Bioassay Effector Cell FcγRIIa (R variant) -NFAT |
| Product Type | Reporter Cell |
| Culture Properties | suspension |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | RPMI-1640+10%FBS+1μg/ml puromycin+400μg/ml Hygromycin B |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
ADCP (Antibody-Dependent Cellular Phagocytosis) is a mechanism of antibody-mediated cellular phagocytosis. It serves as an immune elimination mechanism in which monoclonal antibodies (mAbs) target tumor cells, thereby facilitating their clearance from the body by phagocytic immune cells. ADCP is mediated by monocytes, macrophages, neutrophils, and dendritic cells through the expression of FcγRIIa (CD32a), FcγRI (CD64), and FcγRIIIa (CD16a), which enable the phagocytosis of diseased cells; studies have indicated that FcγRIIa is the primary FcγR receptor involved in this process.
Traditional methods for assaying ADCC/ADCP primarily rely on the isolation and *in vitro* differentiation of relevant primary immune cells, followed by the measurement of target cell killing or phagocytosis effects. These methods are heavily dependent on primary cells derived from human donors; they are expensive, time-consuming, and labor-intensive. Furthermore, the isolation and differentiation of primary cells impose extremely stringent requirements on experimental technique, leading to a significant risk of experimental failure. Moreover, issues regarding the purity of isolated and differentiated cells often result in low detection signals as well as instability and lack of uniformity in the assay results. Additionally, primary cells are a non-renewable resource; since the cell donor source may vary from one experiment to the next, the reproducibility of experimental results is often poor, and batch-to-batch variability can be a significant issue. Due to the influence of these factors, it remains challenging to establish stable and reliable assay methods within the context of drug development, where high-quality control standards are strictly required.
The Jurkat E6.1 Human ADCP Bioassay Effector Cell FcγRIIa (R variant) -NFAT target model effectively mimics the in vivo ADCP signaling transduction process. The principle is illustrated in the figure below.
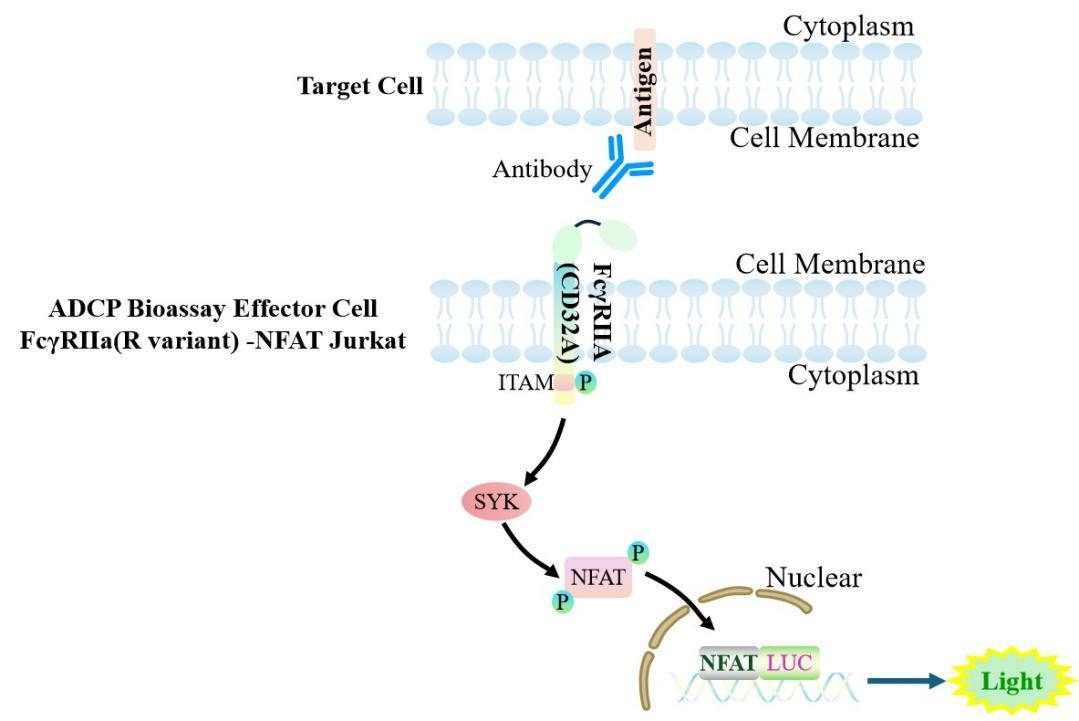
Figure 1. Schematic diagram of the Jurkat E6.1 Human ADCP Bioassay Effector Cell FcγRIIa (R variant) -NFAT cell model.
| Classification | Fc Effector |
| Family | Fc receptor family (FcγR family) |
| Gene Name | FCGR2A |
| Gene Aliases | FCG2;FCGR2A1;FCGR2;CD32;CD32A;IGFR2;CDw32;Fc-gamma-RIIa;FcgammaRIIa |
| Gene ID | 2212 |
| Accession Number | NM_001136219.3 |
| UniProt Number | P12318 |
| Protein Name | IgG Fc receptor II-a |
| Protein Aliases | CDw32;Fc-gamma RII-a (Fc-gamma-RIIa; FcRII-a) |
| Target Species | Human |
| Host cell | Jurkat E6.1 |
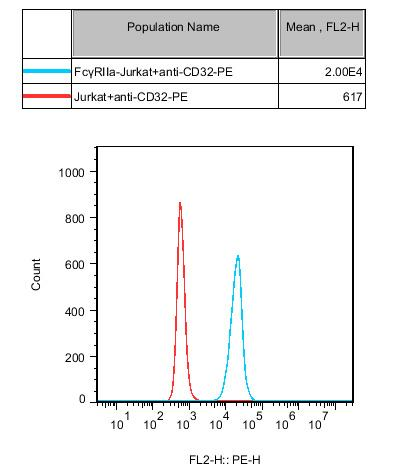
Figure 2. ADCP Bioassay Effector Cell FcγRIIa (R variant) -NFAT Jurkat Cell Flow Cytometry Verification of FcγRⅡa Expression.

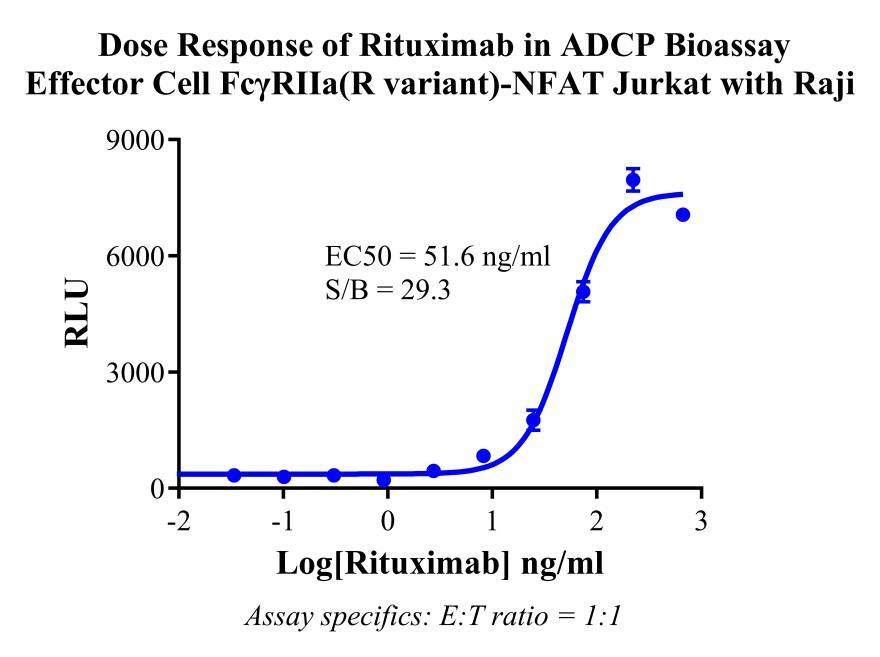
Figure 3. Dose Response of Rituximab in ADCP Bioassay Effector Cell FcγRIIa(R variant)-NFAT Jurkat with Raji.
Cell Passage Procedures
1.This cell line grows in suspension.
2.Upon receipt, cells should be thawed immediately or stored in liquid nitrogen until use.
3.Before thawing, pre-warm the water bath and culture medium to 37 °C, and prepare a small amount of dry ice.
4.Remove the cryovial from storage and transport it to the cell culture laboratory on dry ice.
5.Rapidly thaw the cells in a 37 °C water bath. Once the cells are completely thawed, spray the cryovial with 70% ethanol for disinfection and transfer it to a biosafety cabinet.
6.Add 10 mL of pre-warmed culture medium into a 15 mL centrifuge tube. Transfer the contents of the cryovial into the tube and centrifuge at 1000 rpm for 5 minutes.
7.Carefully discard the supernatant. Resuspend the cell pellet in 5 mL of pre-warmed culture medium by gentle pipetting. Immediately perform cell counting and adjust the cell density to 3–6 × 10⁵ cells/mL based on the counting results, then transfer the cells into a culture flask.
8.Count the cells every 1–2 days. When the cell density exceeds 1 × 10⁶ cells/mL, passage the cells promptly or add fresh culture medium. Maintain the cell density between 2 × 10⁵ and 1 × 10⁶ cells/mL.
Suspension Cell Cryopreservation Procedure:
1.Collect 8 × 10⁶ cells, centrifuge, and discard the supernatant.
2.Add 1 mL of cell freezing medium (90% FBS + 10% DMSO) and gently pipette to mix thoroughly. Transfer the suspension into a cryovial.
3.Immediately place the cryovial into a controlled-rate freezing container (Nalgene 5100-0001), fill with isopropanol up to the indicated level, and store at −80 °C.
4.After 24 hours, transfer the cryovial to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore