
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
HEK293 Human NOD2 Effector Reporter Cell
Cat. No: RQP74536
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74536 |
| Product Name | HEK293 Human NOD2 Effector Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | Adherent |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | MEM +10%FBS + 1% NEAA+ 1mM NaP+1 μg/ml Puromycin+ 100 μg/ml Hygromycin B |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
| Classification | TLR |
| Family | NOD1-NOD2 family |
| Gene Name | NOD2 |
| Gene Aliases | IBD1;CARD15;BLAU;CD;PSORAS1;CLR16.3;NLRC2 |
| Gene ID | 64127 |
| Accession Number | NM_001370466.1 |
| UniProt Number | Q9HC29 |
| Protein Name | Nucleotide-binding oligomerization domain-containing protein 2 |
| Protein Aliases | Caspase recruitment domain-containing protein 15;Inflammatory bowel disease protein 1 |
| Target Species | Human |
| Host cell | HEK293 |

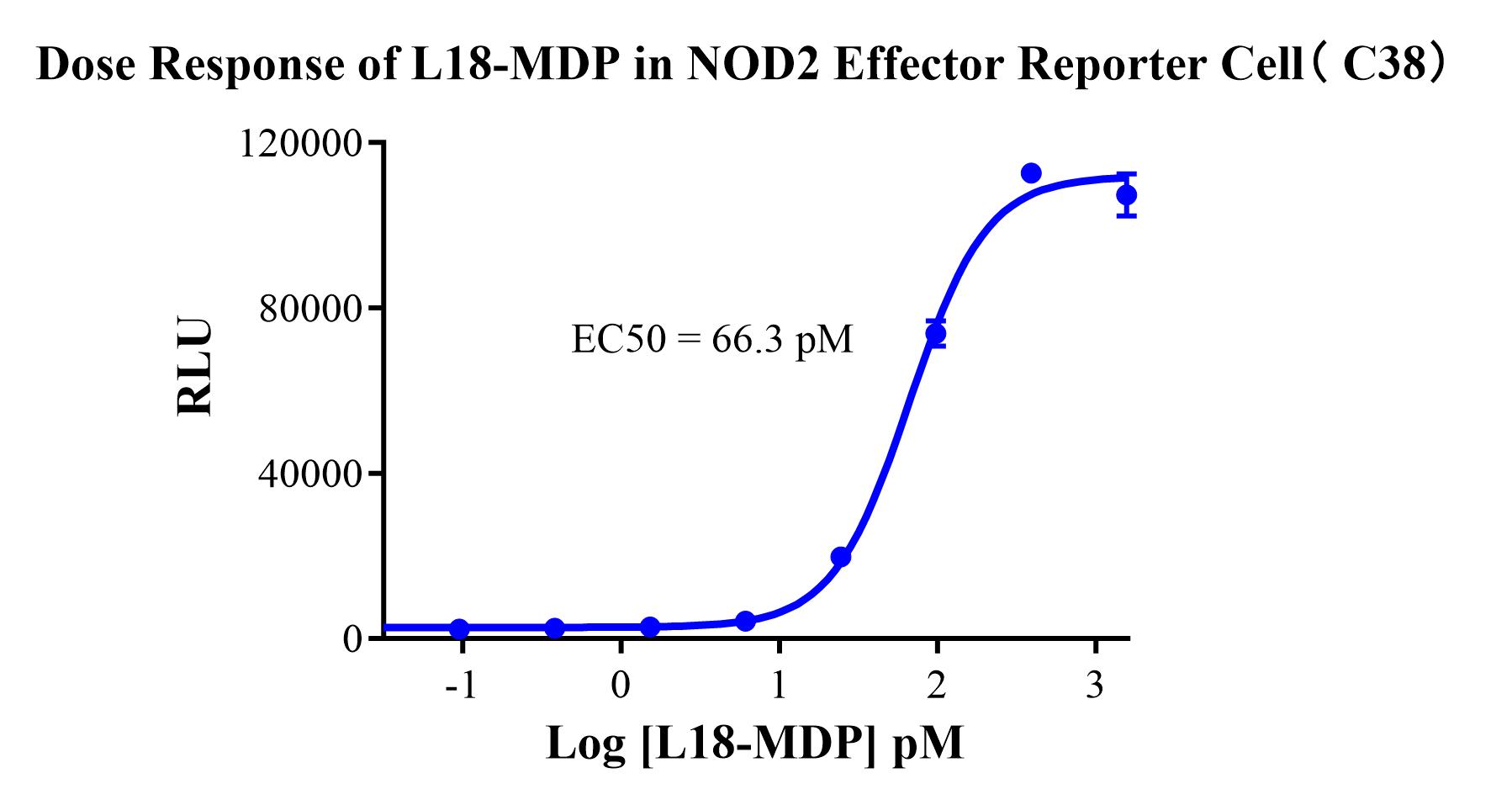
Figure 1. Dose Response of L18-MDP in NOD2 Effector Reporter Cell(C38).
Cell Resuscitation
1)Rapidly thaw the frozen cells in a 37 °C water bath for approximately 60 seconds. Once thawed (which may take slightly less or more than 60 seconds), immediately transfer the cell suspension from the cryovial into a 15 mL centrifuge tube containing 10 mL of pre-warmed HEK293 Human NOD2 Effector Reporter Cell complete culture medium.
2)Centrifuge cells at 1000 rpm for 5 min to remove medium, then resuspend cells in 5 mL of pre-warmed complete medium.
3)Transfer the cell suspension into a T25 culture flask and incubate at 37 °C with 5% CO₂.
4)After approximately 24–36 hours, replace the medium or passage the cells to remove non-adherent dead cells.
Subculturing procedure
1)When the cell density reaches the appropriate confluency for passaging, wash the cells with PBS, then add 1 mL trypsin to detach the cells. When more than 80% of the cells detach upon gently tapping the culture flask, add complete culture medium to terminate digestion. Gently pipette to obtain a single-cell suspension, transfer to a 15 mL centrifuge tube, and centrifuge at 1000 rpm for 5 minutes.
2)Discard supernatant after centrifugation. Resuspend cells in fresh medium to a single-cell suspension and transfer to a new culture flask for continued growth.
Cell Freezing
After trypsinization and centrifugation of cells from each T75 flask or 10 cm culture dish, discard the supernatant. Add 2 mL of cryopreservation medium (90% FBS + 10% DMSO), gently resuspend thoroughly, and aliquot into two cryovials. Immediately place the cryovials into a controlled-rate freezing container (e.g., Nalgene 5100-0001), fill with isopropanol to the indicated level, and store at −80 °C. After 24 hours, transfer the cryovials to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore