
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
HEK293 Human FGFR2b Effector Reporter Cell
Cat. No: RQP74196
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Biological Information
Assay Data
Cell Culture
| Cat. No | RQP74196 |
| Product Name | HEK293 Human FGFR2b Effector Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | Adherent |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | DMEM+10%FBS+100μg/ml Hygromycin B+2μg/ml puromycin |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
| Application | Functional(Report Gene) Assay |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
FGFR2B is one of the splicing variants of fibroblast growth factor receptor 2 (FGFR2) and belongs to the receptor tyrosine kinase (RTK) family. It consists of an extracellular ligand-binding domain (containing three immunoglobulin-like domains), a transmembrane region, and an intracellular tyrosine kinase domain. Its specificity is determined by the alternative splicing of exon IIIb. It is primarily expressed in epithelial cells and forms a paracrine signaling axis with the stromal cell-derived FGFR2C (IIIc splice variant), regulating epithelial-stromal interactions.
The ligands for FGFR2B are FGF7 (KGF), FGF10, and FGF22, which bind to its extracellular domain to induce receptor dimerization and autophosphorylation, thereby activating downstream MAPK/ERK, PI3K-AKT, and STAT signaling pathways to drive cell proliferation, survival, migration, and differentiation. Therapeutic strategies targeting FGFR2B include monoclonal antibodies (such as bemarituzumab, which targets the FGFR2B IIIb epitope and is used in combination with chemotherapy in phase III clinical trials for gastric cancer), small-molecule pan-FGFR inhibitors (erdafitinib, pemigatinib), and selective FGFR2B inhibitors (e.g., LY3076226); however, challenges include overcoming resistance (e.g., the FGFR2 V564F gatekeeper mutation) and off-target toxicity (e.g., hyperphosphatemia). Recent research has focused on combining these agents with immunotherapy (such as anti-PD-1 antibodies) or developing bispecific antibodies to enhance antitumor effects and suppress microenvironment-mediated resistance.
The FGFR2b Effector Reporter Cell model effectively mimics the in vivo signaling pathway of FGFR2b; the underlying mechanism is illustrated in the figure.
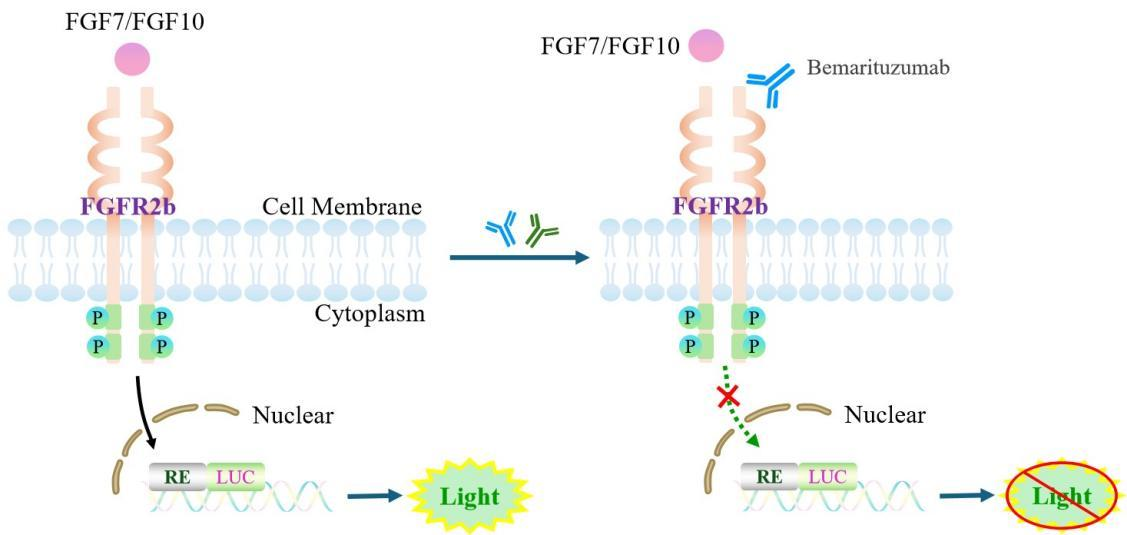
Figure 1. Schematic diagram of the FGFR2b Effector Reporter Cell model
| Classification | Cytokine&Growth Factor |
| Family | Receptor tyrosine kinase (RTK) family |
| Gene Name | FGFR2 |
| Gene Aliases | KGFR;BEK;CFD1;JWS;CEK3;TK14;TK25;ECT1;K-SAM;CD332 |
| Gene ID | 2263 |
| Accession Number | NM_000141.5 |
| UniProt Number | P21802 |
| Protein Name | FGFR-2 |
| Protein Aliases | K-sam (KGFR);Keratinocyte growth factor receptor |
| Target Species | Human |
| Host cell | HEK293 |
https://www.cobioer.com/vancheerfile/Images/2026/3/2026032409439921.jpg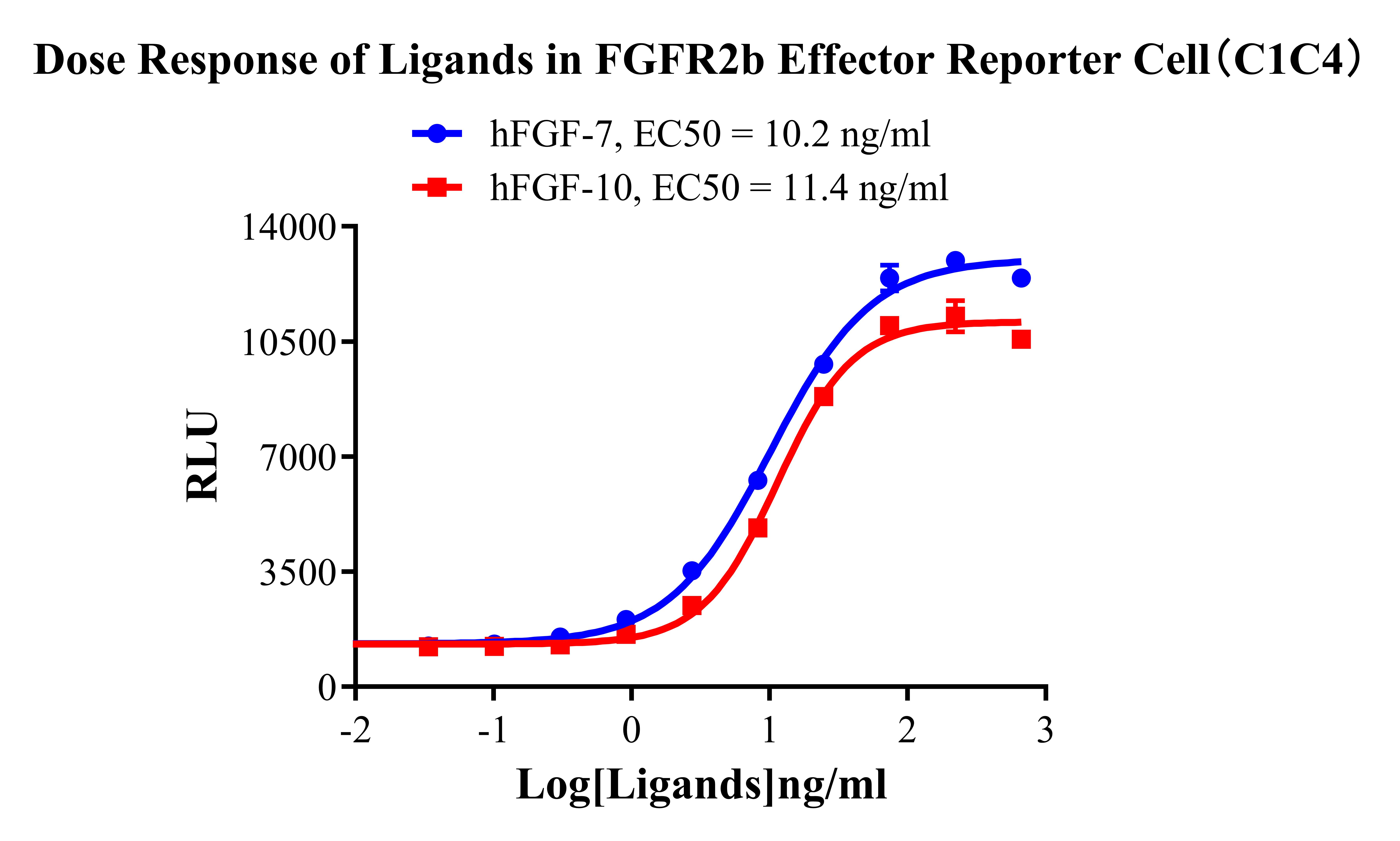
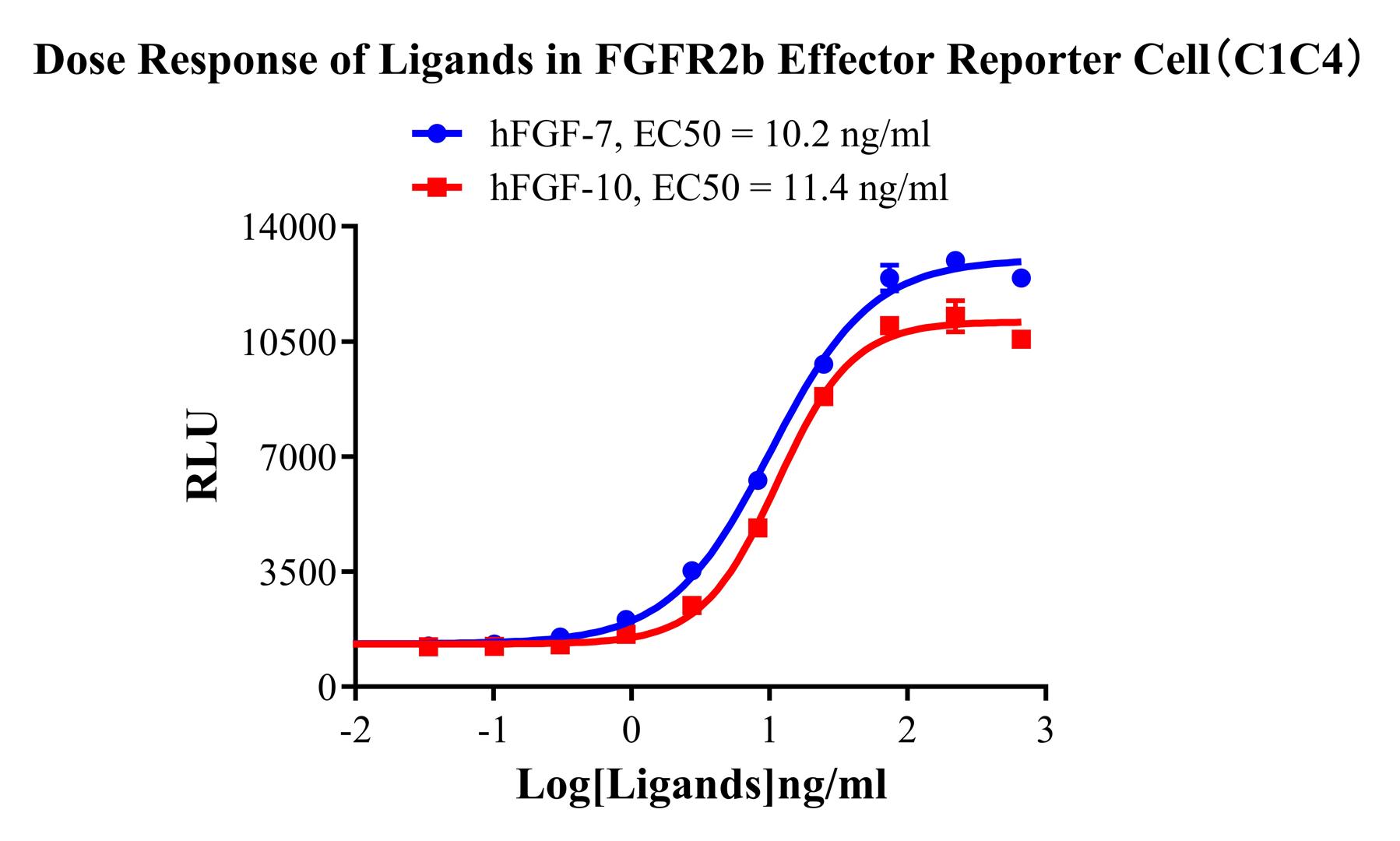
Figure 2. Dose Response of Ligands in FGFR2b Effector Reporter Cell (C1C4).

Figure 3. Inhibition of FGF-7-Induced Reporter Activity by FGFR2b Blocking Ab in FGFR2b Effector Reporter Cell (C1C4).Inhibition of FGF-10-Induced Reporter Activity by FGFR2b Blocking Ab in FGFR2b Effector Reporter Cell (C1C4).
Cell Resuscitation
1)Rapidly thaw the frozen cells in a 37 °C water bath for approximately 60 seconds. Once thawed (which may take slightly less or more than 60 seconds), immediately transfer the cell suspension from the cryovial into a 15 mL centrifuge tube containing 10 mL of pre-warmed HEK293 Human FGFR2b Effector Reporter Cell complete culture medium.
2)Centrifuge cells at 1000 rpm for 5 min to remove medium, then resuspend cells in 5 mL of pre-warmed complete medium.
3)Transfer the cell suspension into a T25 culture flask and incubate at 37 °C with 5% CO₂.
4)After approximately 24–36 hours, replace the medium or passage the cells to remove non-adherent dead cells.
Subculturing procedure
1)When the cell density reaches the appropriate confluency for passaging, wash the cells with PBS, then add 1 mL trypsin to detach the cells. When more than 80% of the cells detach upon gently tapping the culture flask, add complete culture medium to terminate digestion. Gently pipette to obtain a single-cell suspension, transfer to a 15 mL centrifuge tube, and centrifuge at 1000 rpm for 5 minutes.
2)Discard supernatant after centrifugation. Resuspend cells in fresh medium to a single-cell suspension and transfer to a new culture flask for continued growth.
Cell Freezing
After trypsinization and centrifugation of cells from each T75 flask or 10 cm culture dish, discard the supernatant. Add 2 mL of cryopreservation medium (90% FBS + 10% DMSO), gently resuspend thoroughly, and aliquot into two cryovials. Immediately place the cryovials into a controlled-rate freezing container (e.g., Nalgene 5100-0001), fill with isopropanol to the indicated level, and store at −80 °C. After 24 hours, transfer the cryovials to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore