
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Committed to providing comprehensive support for global drug development
HEK293 Human ARE-Luc(Nrf2 Antioxidant Pathway) Reporter Cell
Cat. No: RQPB0011
Size: 1 vial of frozen cells (>1E6 per vial in 1 mL)
Unit Price: Contact For Pricing
Product Info
Description
Assay Data
Cell Culture
| Cat. No | RQPB0011 |
| Product Name | HEK293 Human ARE-Luc(Nrf2 Antioxidant Pathway) Reporter Cell |
| Product Type | Reporter Cell |
| Culture Properties | Adherent |
| Stability | 32passages (in-house test, that not means the cell line will be instable beyond the passages we tested.) |
| Mycoplasma Status | Negative |
| Culture Medium | DMEM+10%FBS+100ug/ml Hygromycin B |
| Freeze Medium | 90% FBS+10% DMSO |
| Storage Conditions | Liquid nitrogen immediately upon delivery |
For research use only. Not intended for human or animal clinical trials, therapeutic or diagnostic use.
Oxidative stress plays a pivotal role in maintaining the balance between intracellular pro-oxidant and anti-oxidant processes. It is implicated in various human diseases, including chronic obstructive pulmonary disease (COPD), Parkinson's disease (PD), Alzheimer's disease (AD), cancer, and diabetes. Nrf2 (Nuclear factor E2-related factor 2) is a central transcription factor that regulates the cellular defense system against oxidative stress; by binding to antioxidant response elements (AREs), it activates the expression of downstream genes to maintain intracellular redox homeostasis, facilitate detoxification, and exert anti-inflammatory effects. Its signaling pathway plays a critical role in the pathogenesis of cancer, neurodegenerative diseases, and metabolic disorders. Substantial evidence indicates that activating the Nrf2 pathway can mitigate inflammatory damage induced by oxidative stress and inhibit the initiation of early-stage tumors. Consequently, Nrf2 activators represent a promising therapeutic option for inflammatory conditions such as lung injury, liver injury, kidney injury, COPD, PD, and AD.
The Nrf2/ARE signaling pathway serves as the primary mechanism for maintaining intracellular redox homeostasis. Under normal physiological conditions, Nrf2 resides in the cytoplasm; anchored to the cytoplasmic actin cytoskeleton via its association with Keap1—forming a Keap1-Nrf2 complex—it undergoes ubiquitin-mediated degradation facilitated by E3 ubiquitin ligases, thereby maintaining a basal steady-state level of Nrf2 activity. Under conditions of oxidative or electrophilic stress, Nrf2 is released from Keap1 and translocates into the nucleus, where it forms heterodimers with small Maf (sMaf) proteins. These heterodimers subsequently bind to AREs located within the promoter regions of cytoprotective genes, thereby triggering the transcription of Nrf2 target genes. In addition to Keap1-dependent regulation, Nrf2 can also undergo phosphorylation by Glycogen Synthase Kinase-3 (GSK-3) via its Neh6 domain; this phosphorylation event leads to its ubiquitination—mediated by the β-transducin repeat-containing protein (β-TrCP)—and subsequent proteasomal degradation.
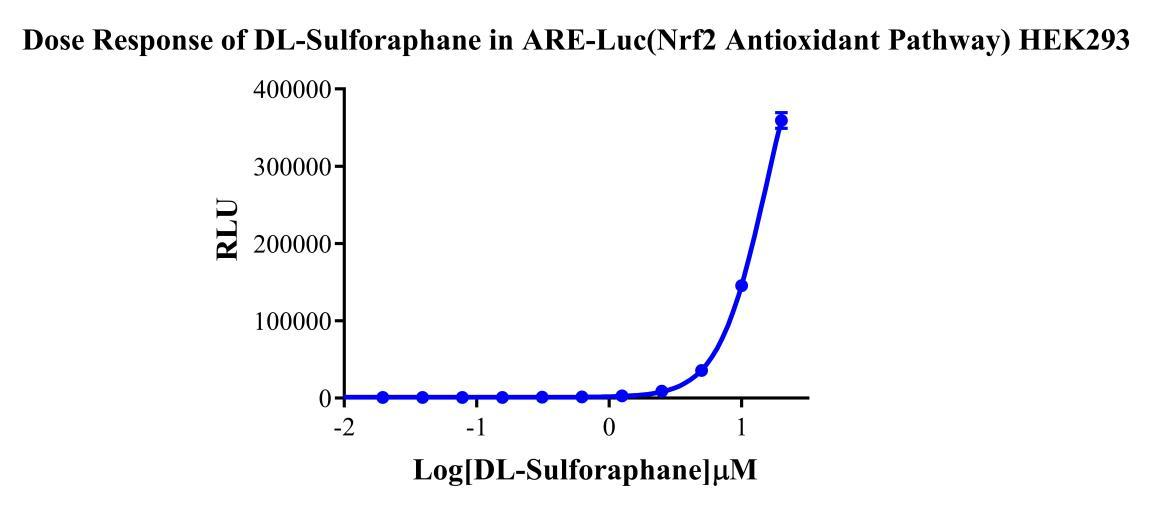
HEK293 Human ARE-Luc(Nrf2 Antioxidant Pathway) Reporter Cells are HEK293 cells in which the Luc luciferase reporter gene is regulated by ARE. Sulforaphane acts as an inducer of the Keap1/Nrf2/ARE pathway; The principle is illustrated in the figure below.

Figure 1. Schematic diagram of the HEK293 Human ARE-Luc(Nrf2 Antioxidant Pathway) Cell Model

Figure 2. Dose response of DL-Sulforaphane in ARE-Luc(Nrf2 Antioxidant Pathway) HEK293.
Cell Resuscitation
1)Rapidly thaw the frozen cells in a 37 °C water bath for approximately 60 seconds. Once thawed (which may take slightly less or more than 60 seconds), immediately transfer the cell suspension from the cryovial into a 15 mL centrifuge tube containing 10 mL of pre-warmed HEK293 Human ARE-Luc(Nrf2 Antioxidant Pathway) Reporter Cell complete culture medium.
2)Centrifuge cells at 1000 rpm for 5 min to remove medium, then resuspend cells in 5 mL of pre-warmed complete medium.
3)Transfer the cell suspension into a T25 culture flask and incubate at 37 °C with 5% CO₂.
4)After approximately 24–36 hours, replace the medium or passage the cells to remove non-adherent dead cells.
Subculturing procedure
1)When the cell density reaches the appropriate confluency for passaging, wash the cells with PBS, then add 1 mL trypsin to detach the cells. When more than 80% of the cells detach upon gently tapping the culture flask, add complete culture medium to terminate digestion. Gently pipette to obtain a single-cell suspension, transfer to a 15 mL centrifuge tube, and centrifuge at 1000 rpm for 5 minutes.
2)Discard supernatant after centrifugation. Resuspend cells in fresh medium to a single-cell suspension and transfer to a new culture flask for continued growth.
Cell Freezing
After trypsinization and centrifugation of cells from each T75 flask or 10 cm culture dish, discard the supernatant. Add 2 mL of cryopreservation medium (90% FBS + 10% DMSO), gently resuspend thoroughly, and aliquot into two cryovials. Immediately place the cryovials into a controlled-rate freezing container (e.g., Nalgene 5100-0001), fill with isopropanol to the indicated level, and store at −80 °C. After 24 hours, transfer the cryovials to liquid nitrogen for long-term storage.
Related products
HEK293 Human NK1R CRE-Luc Cell Line
Raji-Luc-GFP
Jurkat E6.1-Luc
THP-1-GFP
THP-1-Luc
Raji-GFP
Raji-Luc
Jurkat E6.1-GFP
HEK293 Human GAL4-Luc Reporter Cell
HEK293 Human CRE-Luc Reporter Cell
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore