
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Professional technology integration to support the entire R&D process
Home>
Resources>
Learning>
Articles>
Focusing on the IL-17 Target: The "Golden Track" of Autoimmune Drug R&D and Essential Tool Models
Focusing on the IL-17 Target: The "Golden Track" of Autoimmune Drug R&D and Essential Tool Models
In recent years, with the deepening of immunological research, Interleukin-17 (IL-17) has emerged as a pivotal target for treating autoimmune diseases, consistently demonstrating immense clinical value and market potential. Following the successful launch of several blockbuster drugs and the continuous expansion of new indications, IL-17 has become a "Golden Track" for pharmaceutical competition.
To empower drug R&D in this field, ReqBioBio has leveraged cutting-edge technology to develop a series of IL-17 family cell models, providing robust tool support for early-stage mechanism of action (MoA) studies and drug screening.
I. IL-17: More Than a Pro-inflammatory Cytokine, a Multifunctional Immune Regulator
The IL-17 family currently comprises six members (IL-17A to IL-17F; IL-17E is also known as IL-25) and five receptors. As the family's archetype, IL-17A has gained significant attention for its core driving role in diseases like psoriasis and ankylosing spondylitis (AS). However, recent studies indicate that IL-17’s function is far more complex than simple "pro-inflammation"; it also plays a vital role in injury response, physiological stress, and maintaining tissue homeostasis.
IL-17 Signaling Pathway Overview:
IL-17 initiates signal transduction primarily by binding to corresponding receptor complexes (e.g., IL-17RA/IL-17RC). This process involves the activation of Act1 and TRAF family proteins (such as TRAF6), which in turn triggers downstream NF-κB, MAPK, and C/EBP signaling pathways. This ultimately induces the expression of pro-inflammatory cytokines (e.g., IL-6, IL-8) and chemokines, recruiting neutrophils and leading to local inflammation. Modulating this pathway is now a central strategy for treating various autoimmune disorders.
II. A Competitive Landscape: IL-17 Drug Pipeline and Market Outlook
Due to its proven efficacy, the IL-17 target has become a hub for "Blockbuster" drugs in the autoimmune sector.
Marketed Drugs: Six monoclonal antibodies (mAbs) targeting the IL-17 family are currently approved worldwide. Novartis’s Secukinumab, the world’s first IL-17A inhibitor, surpassed $4 billion in sales by 2020. Other key players include Eli Lilly’s Ixekizumab, Amgen’s Brodalumab, and UCB’s Bimekizumab (targeting IL-17A/F).
Indication Expansion: Beyond psoriasis and AS, IL-17 therapies are aggressively expanding into Inflammatory Bowel Disease (IBD), Multiple Sclerosis (MS), and other broader immune-mediated conditions.
R&D Pipeline: Innovation is accelerating. Over 20 drug candidates are currently in clinical stages, diversifying from mAbs to bispecific antibodies and small molecule inhibitors.
IL-17 Drug Development Pipeline:
|
Name |
Developer |
Phase |
Target |
Drug Type |
Indication |
|
Izokibep (IMG-020) |
Inmagene Biopharmaceuticals/Affibody/ACELYRIN |
Phase III |
IL-17A |
Biologic |
Psoriasis, Ankylosing Spondylitis, etc. |
|
AK-111 |
Akeso |
Phase II |
IL-17A |
Biologic |
Plaque Psoriasis |
|
Anti-IL-17A humanized mAb |
Sunshine Guojian |
Phase II |
IL-17A |
Biologic |
Plaque Psoriasis |
|
Anti-IL-17A humanized mAb |
Junshi Biosciences |
Phase II |
IL-17A |
Biologic |
Plaque Psoriasis |
|
HB-0017 |
Huabo Biopharm |
Phase I |
IL-17A |
Biologic |
Moderate-to-Severe Plaque Psoriasis |
|
QX-002N |
Qyuns Therapeutics |
Phase I |
IL-17A |
Biologic |
Ankylosing Spondylitis |
|
ZL-1102 |
Zai Lab |
Phase I |
IL-17A |
Biologic |
Plaque Psoriasis |
|
SIM0335 |
Simcere Pharmaceutical |
Phase I |
IL-17A |
Small molecule |
Plaque Psoriasis |
|
LY3509754 |
Eli Lilly |
Phase I |
IL-17A |
Small molecule |
Autoimmune Disease |
|
ASC50 |
Ascletis |
Phase I |
IL-17A |
Small molecule |
Psoriasis |
(Note: Only representative projects are shown here; data updated to 2024)
III. Empowering R&D: Reqbio Bio’s IL-17 Functional Cell Models
In a highly competitive R&D landscape, efficient and stable early-stage screening tools are the key to gaining a first-mover advantage. ReqBioBio has successfully developed and validated critical cell models targeting the IL-17 family.
1. IL-17A/F Cell Model
Given the homology between IL-17A and IL-17F, and their ability to form homodimers (IL-17A/A, IL-17F/F) or heterodimers (IL-17A/F), models that accurately evaluate different ligand forms are essential.
l Feature: Specifically responds to IL-17 family ligands.
l Application: Uses a reporter gene system to visualize the blocking effect of mAbs, bispecifics, or small molecules on downstream pathways. Ideal for in vitro activity and neutralization assays.
Jurkat E6.1 Human IL17A/17F Effector Reporter Cell (Cat No. RQP74484)
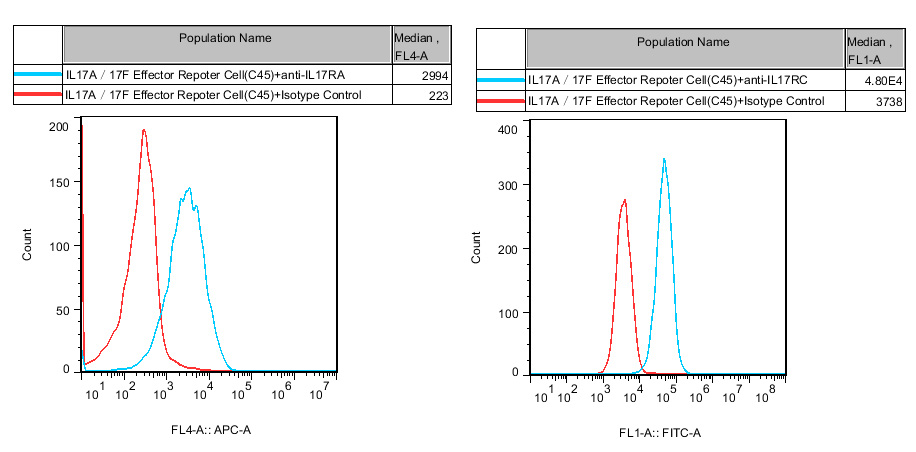
Figure 1.Dose Response of Ligands in IL17A/17F Effector Reporter Cell (C45).
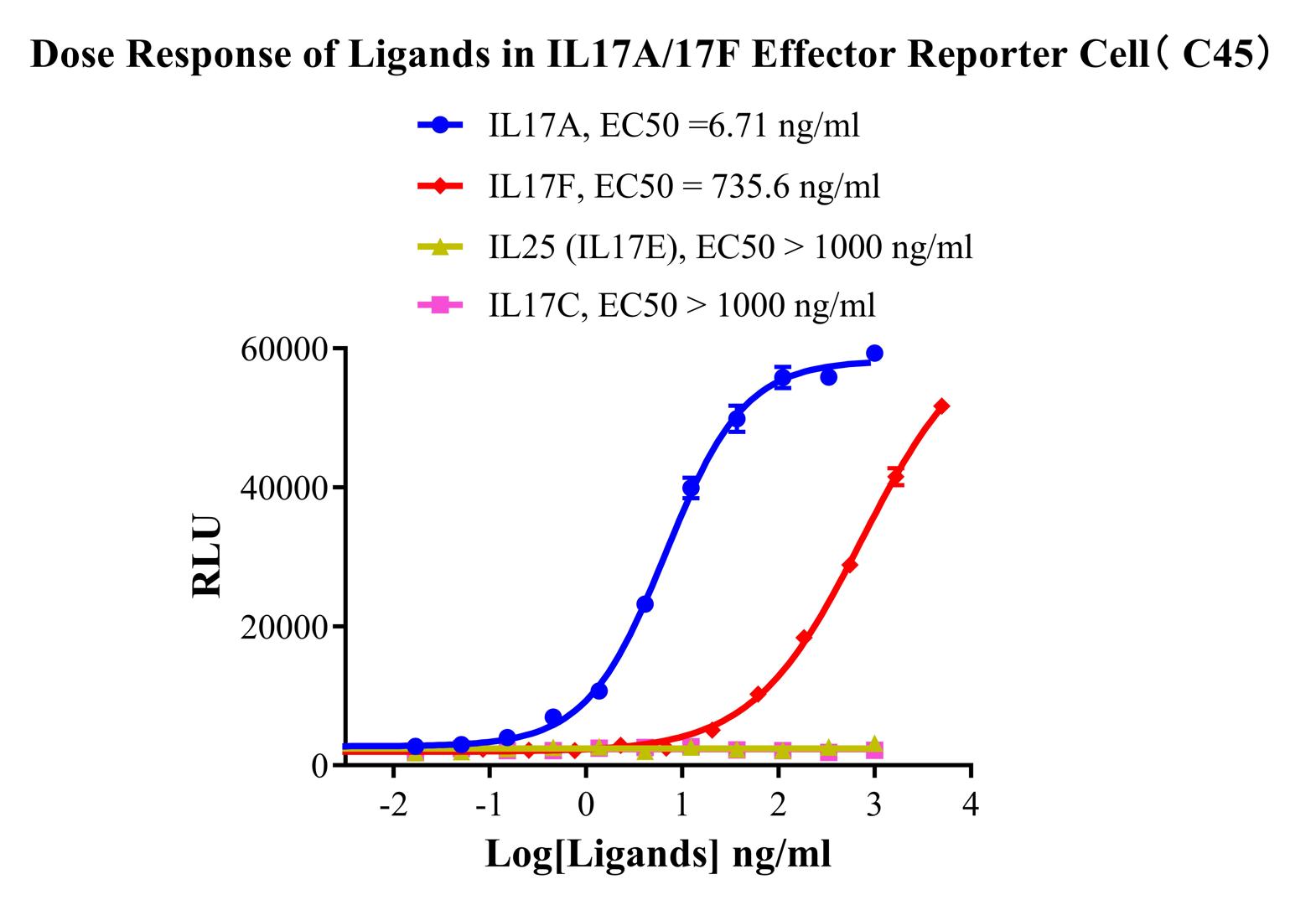
Figure 2. Inhibition of Human IL17A induced Reporter Activity by IL17RA Blocking or IL17A Neutralization Ab in IL17A/17F Effector Reporter Cell(C45).
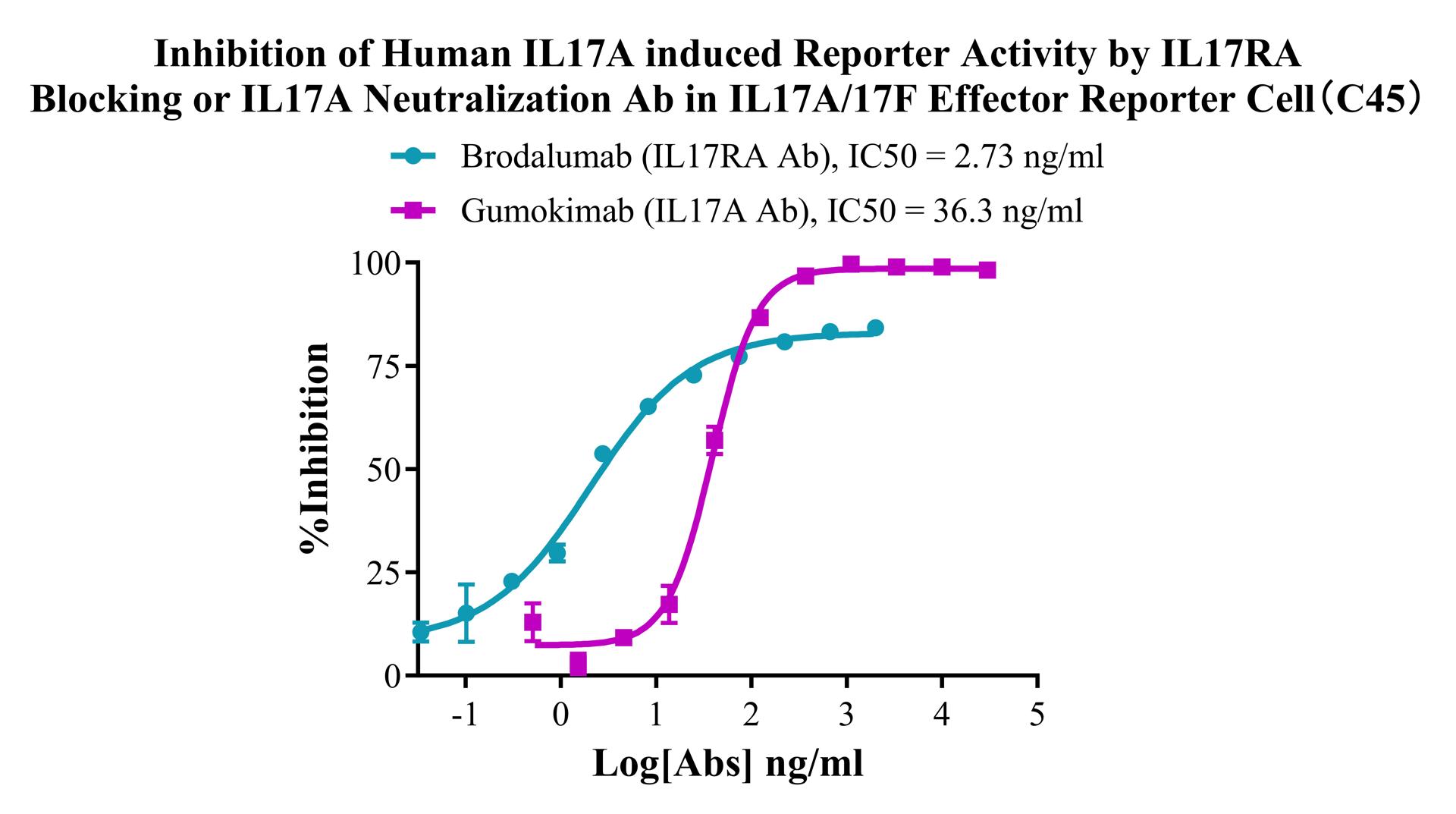
Figure 3. Inhibition of Human IL17A induced Reporter Activity by IL17RA Blocking or IL17A Neutralization Ab in IL17A/17F Effector Reporter Cell(C45).
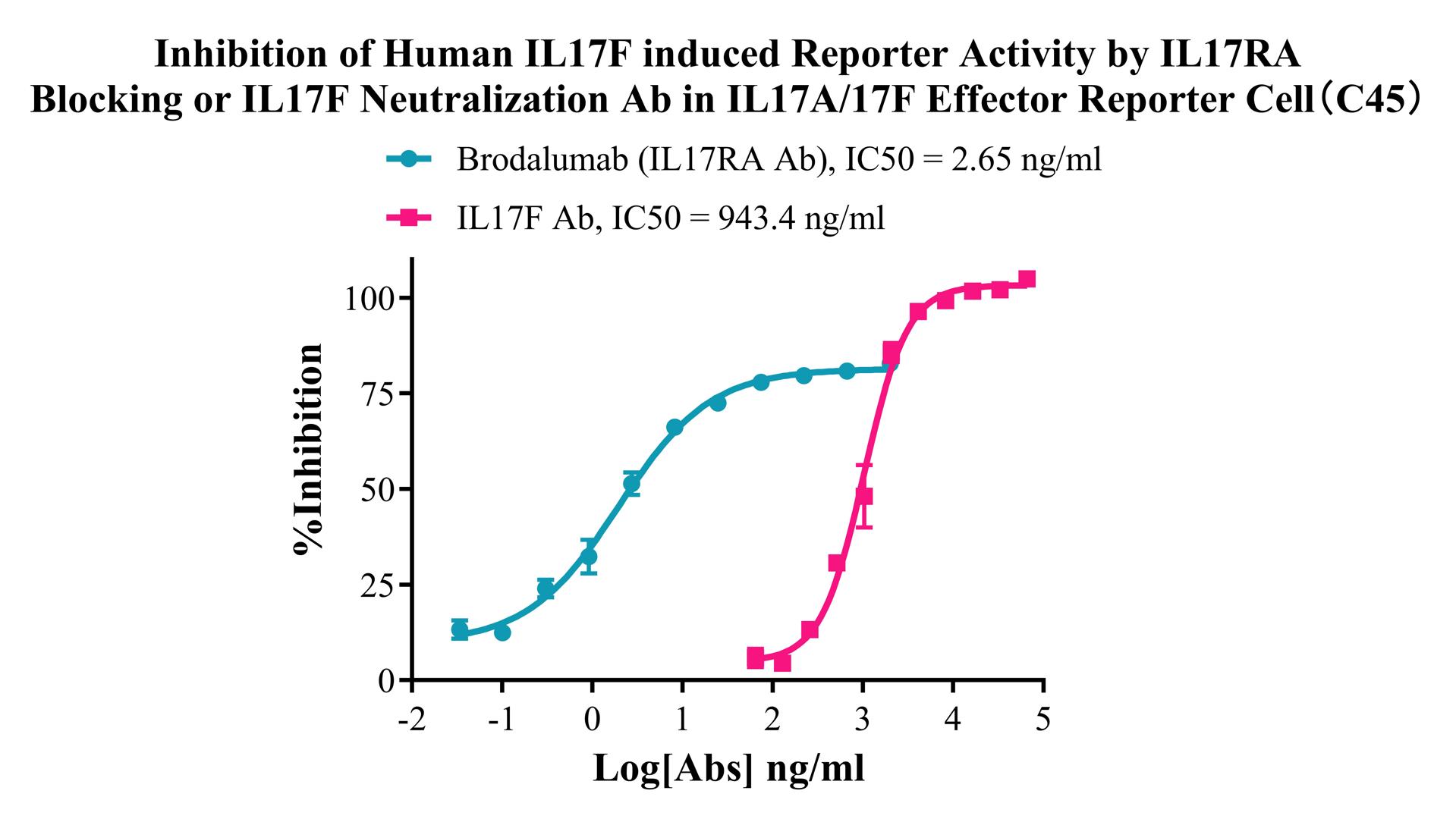
Figure 4. Inhibition of Human IL17F induced Reporter Activity by IL17RA Blocking or IL17F Neutralization Ab in IL17A/17F Effector Reporter Cell(C45).
2. IL-25 (IL-17E) Cell Model
IL-25 is a unique family member involved in Type 2 immune responses, playing a major role in allergic diseases and anti-parasitic infections.
l Feature: Provides a reliable bioactivity assay platform for studying IL-25 specific inhibitors and immune regulation mechanisms.
Jurkat E6.1 Human IL25 Effector Reporter Cell(Suspension) (Cat No. RQP74480)
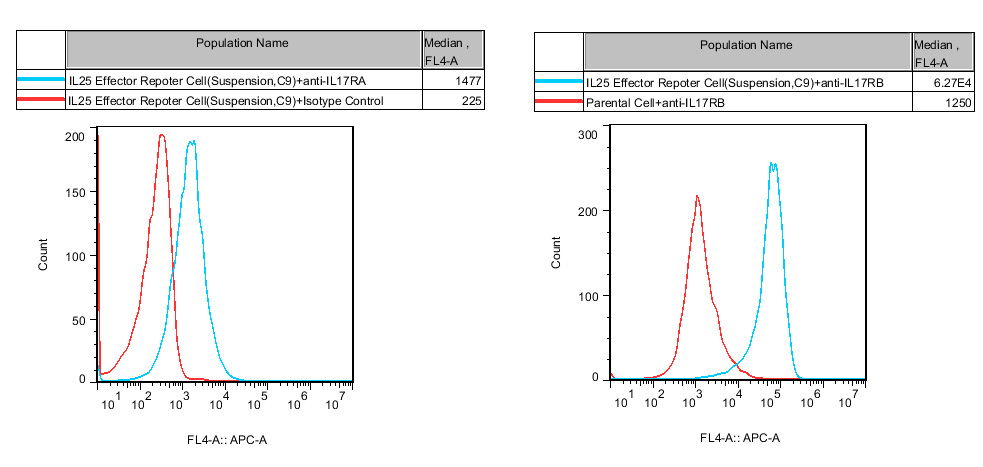
Figure 5. Recombinant IL25 Effector Reporter Cell(Suspension) stably expressing IL17RA & IL17RB.
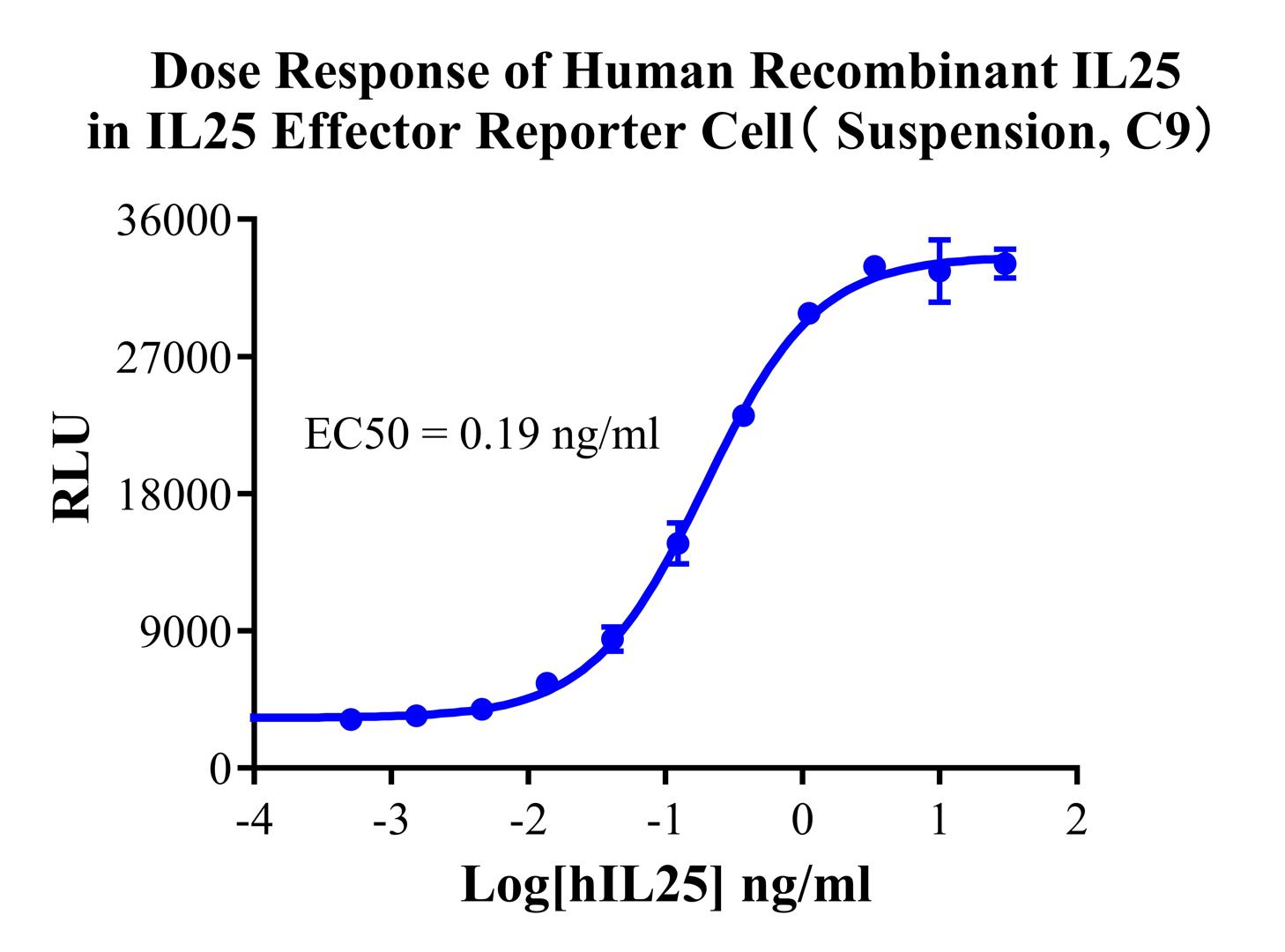
Figure 6. Dose Response of Human Recombinant IL25 in IL25 Effector Reporter Cell(Suspension, C9).
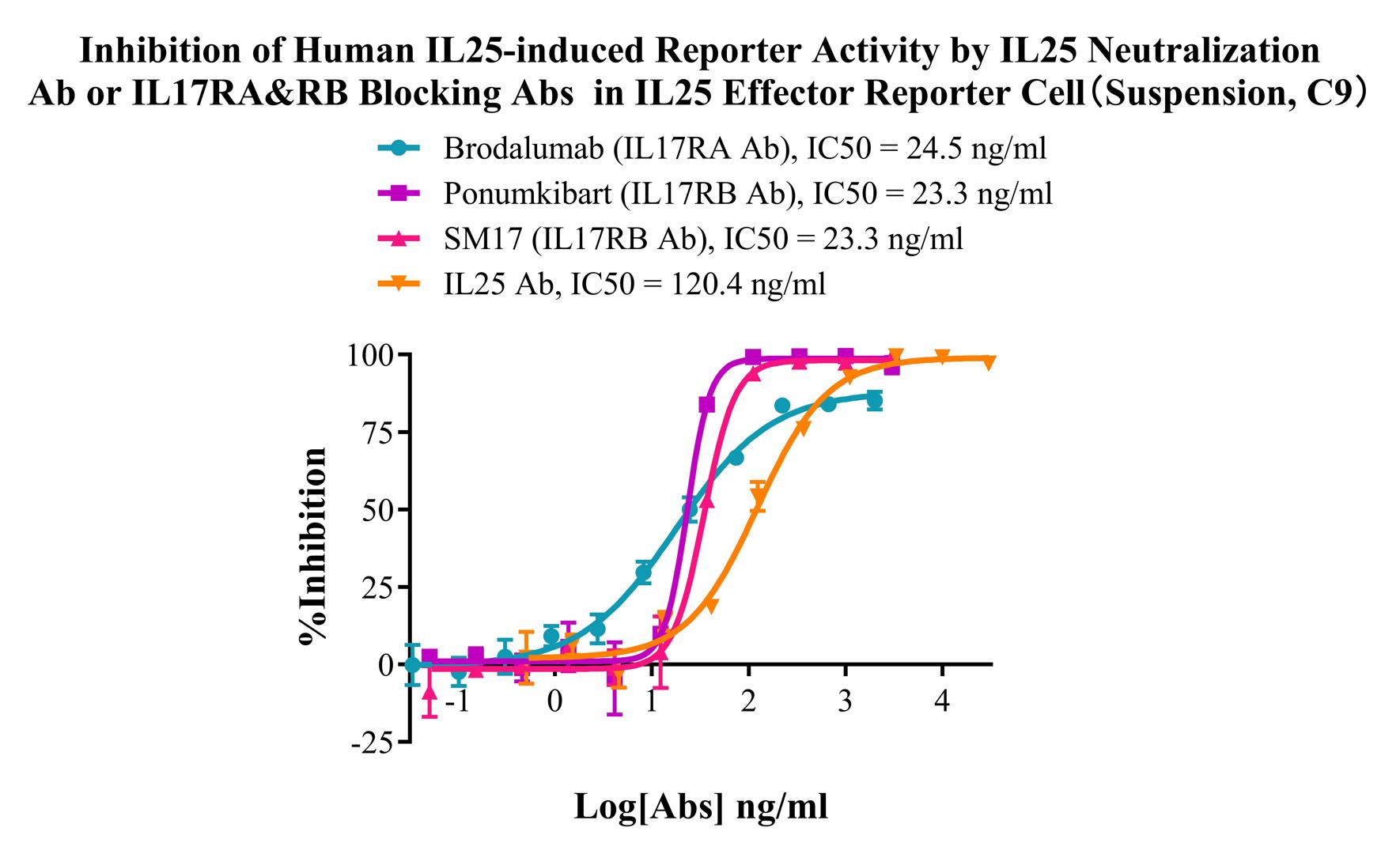
Figure 7. Inhibition of Human IL25-induced Reporter Activity by IL25 Neutralization Ab or IL17RA&RB Blocking Abs in IL25 Effector Reporter Cell (Suspension, C9).
Advantages of ReqBioBio Cell Models:
High Sensitivity & Stability: Ensures reproducible experimental data and reliable screening results.
Clear Mechanism: Rigorous design based on IL-17 signaling pathways to reflect real drug-target interactions.
Ready-to-Use: Shortens R&D cycles and accelerates early drug discovery.
IV. Conclusion
From basic research to clinical translation, the potential of the IL-17 target is expanding exponentially. In an increasingly crowded track, choosing reliable tools means higher efficiency and lower costs. ReqBio remains committed to the frontiers of immunotherapy, providing high-quality biological reagents and cell models to help global researchers conquer autoimmune diseases.
news recommendation
NFAT Signaling Pathway: A Critical Target for Drug R&D and Cell Models
TSHR Cell Model for Thyroid Cancer Drug Screening | GPCR Assay Platform-Reqbio
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore



