
Drug Target Models
GPCR Reporter Cells
Immunotherapy cells
Other Stable Cells
Assay Kits & Reagents
Services
Resources
Company
Professional technology integration to support the entire R&D process
Home>
Resources>
Learning>
Articles>
CHRM1-5 Subtypes: Structure, Mechanism, Drug Discovery, and Cell
CHRM1-5 Subtypes: Structure, Mechanism, Drug Discovery, and Cell
Within the G protein-coupled receptor (GPCR) superfamily, muscarinic acetylcholine receptors (CHRM, or mAChRs) act as the primary conduits for cholinergic signaling. From modulating cognitive function in the central nervous system to maintaining physiological homeostasis in peripheral organs, the five mAChR subtypes (M1–M5) orchestrate a sophisticated regulatory network.
As our understanding of the pathophysiology underlying Alzheimer’s disease, schizophrenia, Chronic Obstructive Pulmonary Disease (COPD), and overactive bladder (OAB) continues to evolve, the distinct therapeutic potential of each CHRM subtype has come into sharp focus. Consequently, they have emerged as high-priority targets in neuropharmacology, respiratory medicine, and cardiovascular research.
Capitalizing on our established GPCR drug discovery platform, ReqBio has successfully engineered a complete suite of stable cell lines covering the M1 through M5 subtypes. By offering functional assays for both the Gq-coupled (IP-One) and Gi-coupled (HTRF cAMP) pathways, we provide global researchers with high-performance, reliable tools to streamline early-stage lead identification and optimization.
I. CHRM1–5: Five Distinct Subtypes with High Homology and Divergent Functions
All five CHRM1–5 subtypes are single-subunit, seven-transmembrane (7TM) glycoproteins belonging to Class A (rhodopsin-like) GPCRs. While their core structures are highly conserved, they are categorized into two distinct functional groups based on their G-protein coupling profiles:
|
Subtype |
Gene Localization |
Primary G Protein |
Signaling Pathway |
Major Distribution |
Core Functions |
|
M1 |
11q13 |
Gq/11 |
PLCβ → IP3/DAG → Ca²⁺/PKC |
Cerebral cortex, Hippocampus, Gastric parietal cells |
Learning and memory, Cognition, Gastric acid secretion |
|
M2 |
7q35-q36 |
Gi/o |
AC Inhibition → ↓cAMP; GIRK activation |
Heart, Brainstem, Presynaptic terminals |
Negative chronotropic effects (heart rate), Presynaptic inhibition |
|
M3 |
1q43 |
Gq/11 |
PLCβ → IP3/DAG → Ca²⁺/PKC |
Exocrine glands, Airways, Bladder, GI smooth muscle |
Glandular secretion, Smooth muscle contraction |
|
M4 |
11p12-p11.2 |
Gi/o |
AC Inhibition → ↓cAMP; β-Arrestin |
Striatum, Nucleus accumbens, Hippocampus |
Locomotor control, Dopaminergic system regulation |
|
M5 |
15q14 |
Gq/11 |
PLCβ → IP3/DAG → Ca²⁺/PKC |
Midbrain dopaminergic neurons, Vascular smooth muscle |
Reward, Addiction, Cerebrovascular regulation |
Table 1: G Protein-Coupled Types
Structure Dictates Function: Despite the high degree of homology across their transmembrane domains, the five subtypes exhibit significant sequence divergence within the third intracellular loop (ICL3). This structural variation is the primary determinant of G-protein coupling specificity. Understanding these differences provides the structural framework for developing subtype-selective ligands, making the screening of highly specific agonists and antagonists achievable.
II. Signal Transduction: Differential Detection of Gq vs. Gi Pathways
Gi-Coupled Subtypes (M2, M4): Activation leads to the inhibition of adenylyl cyclase (AC) via the Gαi subunit, resulting in decreased cAMP levels. Simultaneously, the βγ subunits activate GIRK potassium channels and inhibit calcium channels. The gold standard for assessing these receptors is the HTRF cAMP assay, where agonists are evaluated by their ability to inhibit forskolin-induced cAMP elevation.
Of particular interest is M4, which can also signal through β-arrestin in a G-protein-independent manner. This characteristic adds a new dimension to drug discovery by enabling the development of biased ligands.
III. Targeting CHRM: From Classic Pharmacology to Frontier
TherapeuticsCurrent drug development targeting CHRM primarily focuses on three major therapeutic areas: neurodegenerative diseases, respiratory disorders, and psychiatric conditions. The core R&D strategies center on small-molecule agonists, antagonists, and allosteric modulators.
Representative Marketed Drugs:
Ipratropium & Tiotropium (Primarily M3, with M1/M2 antagonism): Used for bronchodilation in asthma and COPD.
Atropine (Non-selective M1–M5 antagonist): Used to treat bradycardia.
Tolterodine & Solifenacin (M3-selective antagonists): Standard-of-care treatments for Overactive Bladder (OAB).
Pipeline Highlights (Selected):
|
Drug Candidate |
Developer |
Phase |
Target |
Indication |
|
Xanomeline |
Eli Lilly |
Phase III |
M1 & M4 |
Alzheimer’s Disease, Agitation |
|
Direclidine |
Nxera Pharma |
Phase III |
M4 |
Schizophrenia, Alzheimer’s Disease |
|
Velufenacin |
Dong-A Pharmaceutical |
Phase III |
M3 |
Overactive Bladder (OAB) |
|
Emraclidine |
Cerevel Therapeutics |
Phase II |
M4 |
Schizophrenia, Alzheimer’s Disease |
|
NS-136 |
NeuShen Biopharma |
Phase II |
M4 |
Schizophrenia, Alzheimer’s Disease |
|
PIPE-307 |
Contineum Therapeutics |
Phase II |
M1 |
Depression, Multiple Sclerosis (MS) |
|
L-Phencynonate HCl |
Beijing Lansheng Pharma |
Phase II |
M4 |
Parkinson’s Disease |
Table 2: Drugs in the R&D Pipeline
IV. Accelerating CHRM Drug Discovery: ReqBio’s Comprehensive Cell Models and Data Advantages
|
Cell Line Name |
Catalog No. |
Host Cell |
Assay Mode |
G-Protein Coupling |
Core Application |
|
CHO-K1 Human CHRM1 Cell Line |
RQP71180 |
CHO-K1 |
HTRF IP-One |
Gq |
M1 Agonist/Antagonist Screening |
|
CHO-K1 Human CHRM2 Cell Line |
RQP71427 |
CHO-K1 |
HTRF cAMP |
Gi |
M2 Agonist/Antagonist Screening |
|
CHO-K1 Human CHRM3 Cell Line |
RQP71512 |
CHO-K1 |
HTRF IP-One |
Gq |
M3 Agonist/Antagonist Screening |
|
CHO-K1 Human CHRM4 Gα15 Cell Line |
RQP71528 |
CHO-K1 |
HTRF cAMP / IP-One |
Gi(Modification) |
Dual-mode M4 Agonist/Antagonist Screening |
|
CHO-K1 Human CHRM5 Cell Line |
RQP71184 |
CHO-K1 |
HTRF IP-One |
Gq |
M5 Agonist/Antagonist Screening |
Table 3:ReqBio CHRM Series: Stable Cell Line Portfolio
Validation Data & Advantage Analysis
1.CHO-K1 Human CHRM1 Cell Line(RQP71180)
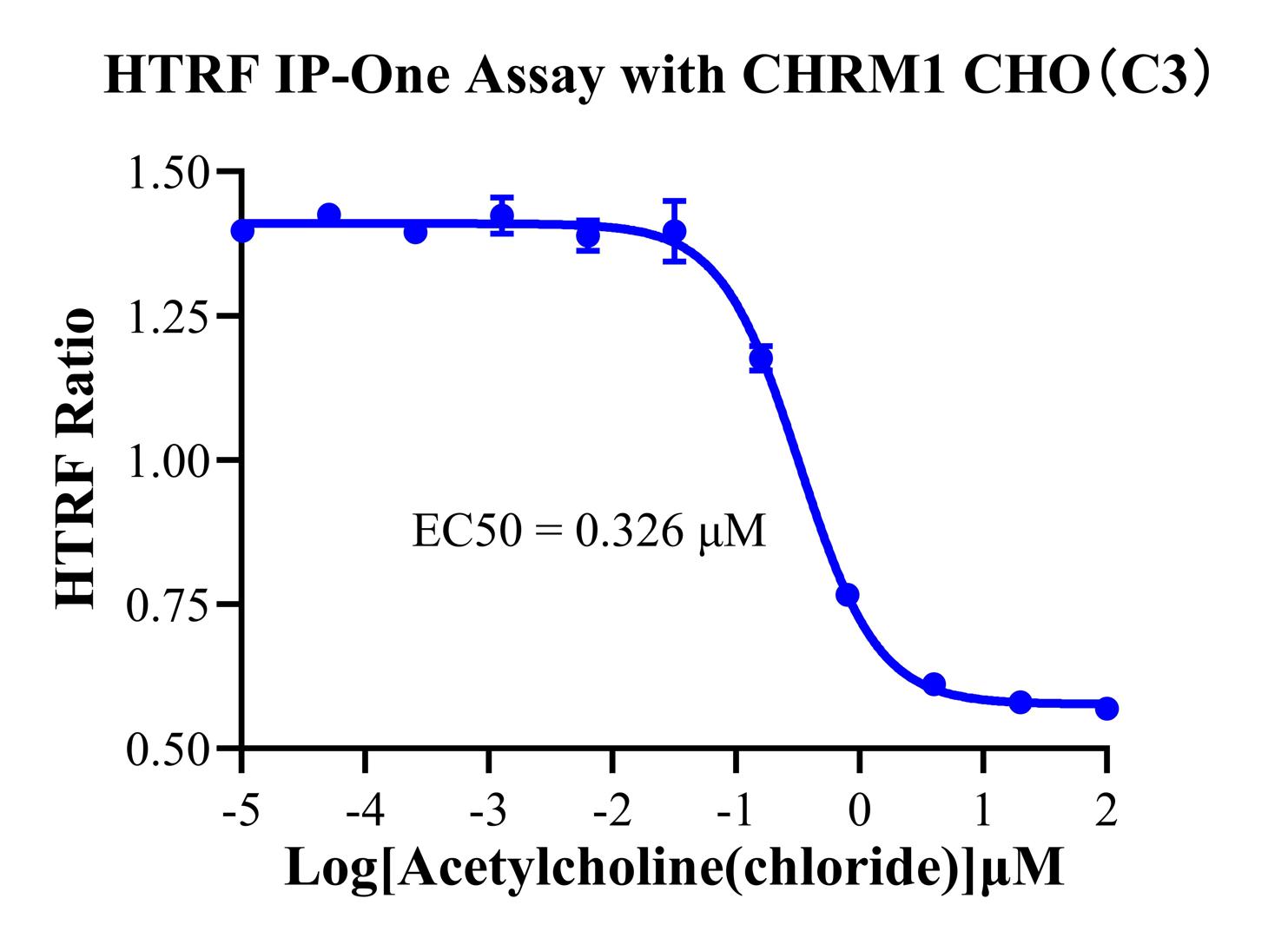
Figure 4. HTRF IP-One Assay with CHRM1 CHO-K1(C3)
Assay Principle: M1 is a Gq-coupled receptor that triggers IP1 accumulation upon activation. We utilize the HTRF IP-One kit, employing a competitive immunoassay format to quantify IP1 levels.
Data Highlights: Figure 4 illustrates a classic dose-response profile (increasing agonist concentration leads to a corresponding rise in the IP1 signal) with a well-defined EC50and a robust signal window (>5-fold). This demonstrates that the cell line is highly sensitive to reference agonists (such as Carbachol), making it an ideal tool for agonist potency ranking and determining the IC50 of prospective antagonists.
2. CHO-K1 Human CHRM2 Cell Line(RQP71427)
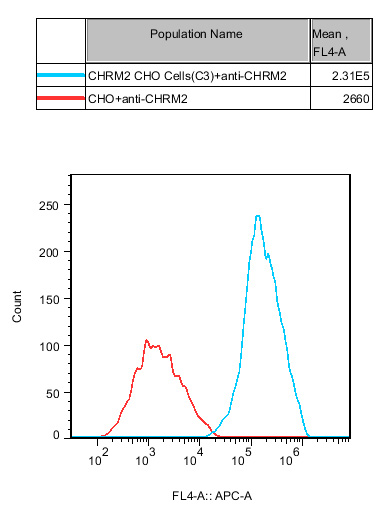
Figure 5: Surface expression or total protein levels of CHRM2 in CHO cells are confirmed via flow cytometry or Western Blot, ensuring high stability and receptor density for model reliability.
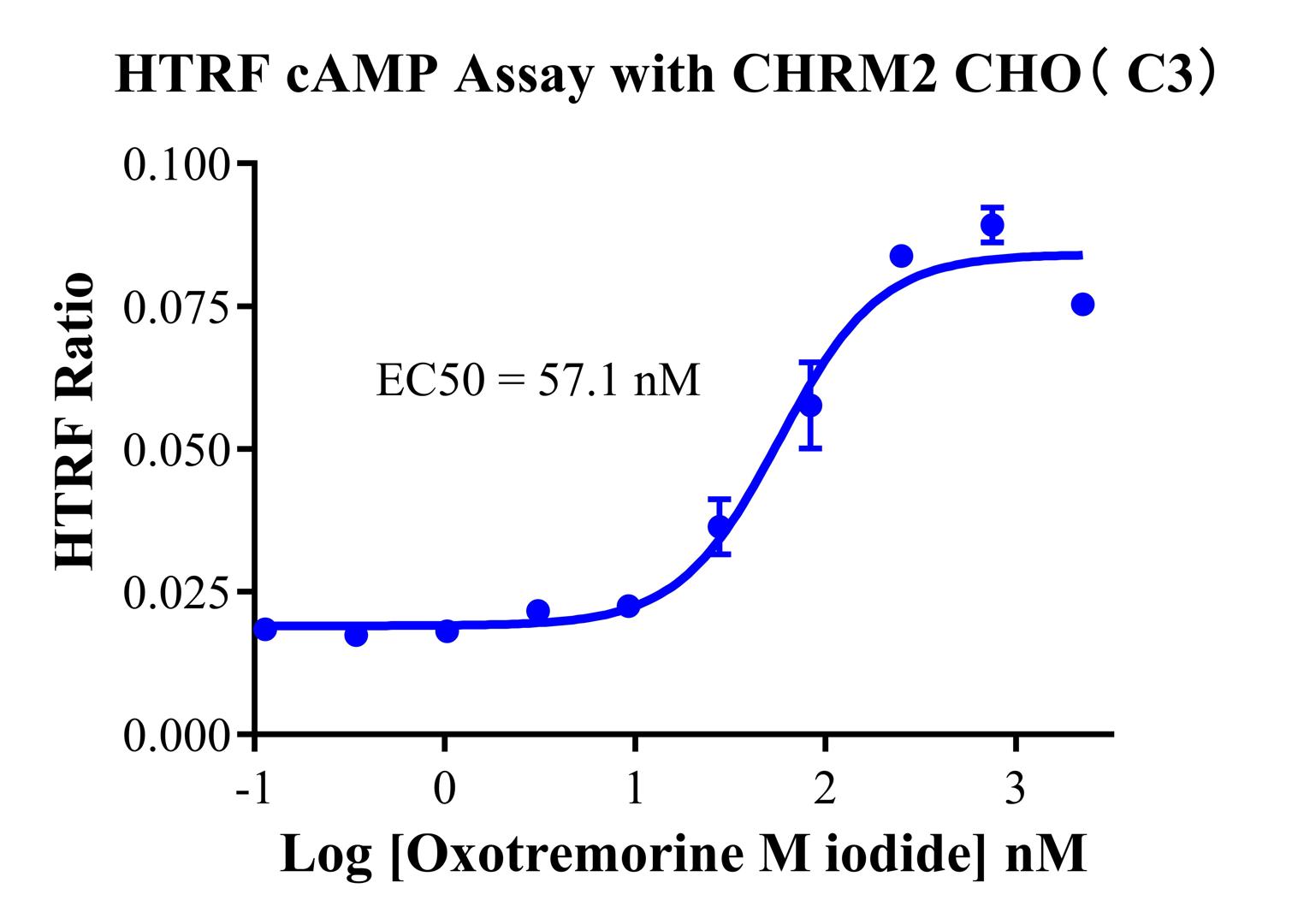
Figure 6: The HTRF cAMP assay demonstrates that M2 agonists induce a dose-dependent inhibition of forskolin-stimulated cAMP levels (characteristic sigmoidal dose-response curve). This model effectively distinguishes between full agonists, partial agonists, and inverse agonists, making it highly suitable for high-throughput screening (HTS) of drugs targeting the Gi pathway.
3. CHO-K1 Human CHRM3 Cell Line(RQP71512)
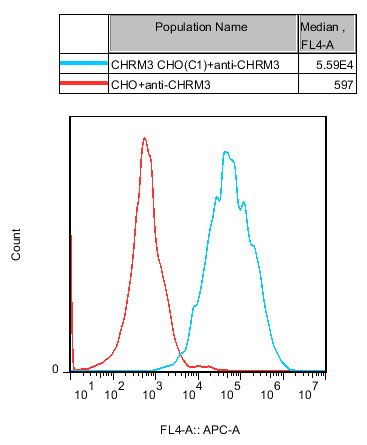
Figure 7: Stable expression of CHRM3 is confirmed, ensuring consistent receptor density across cell generations.
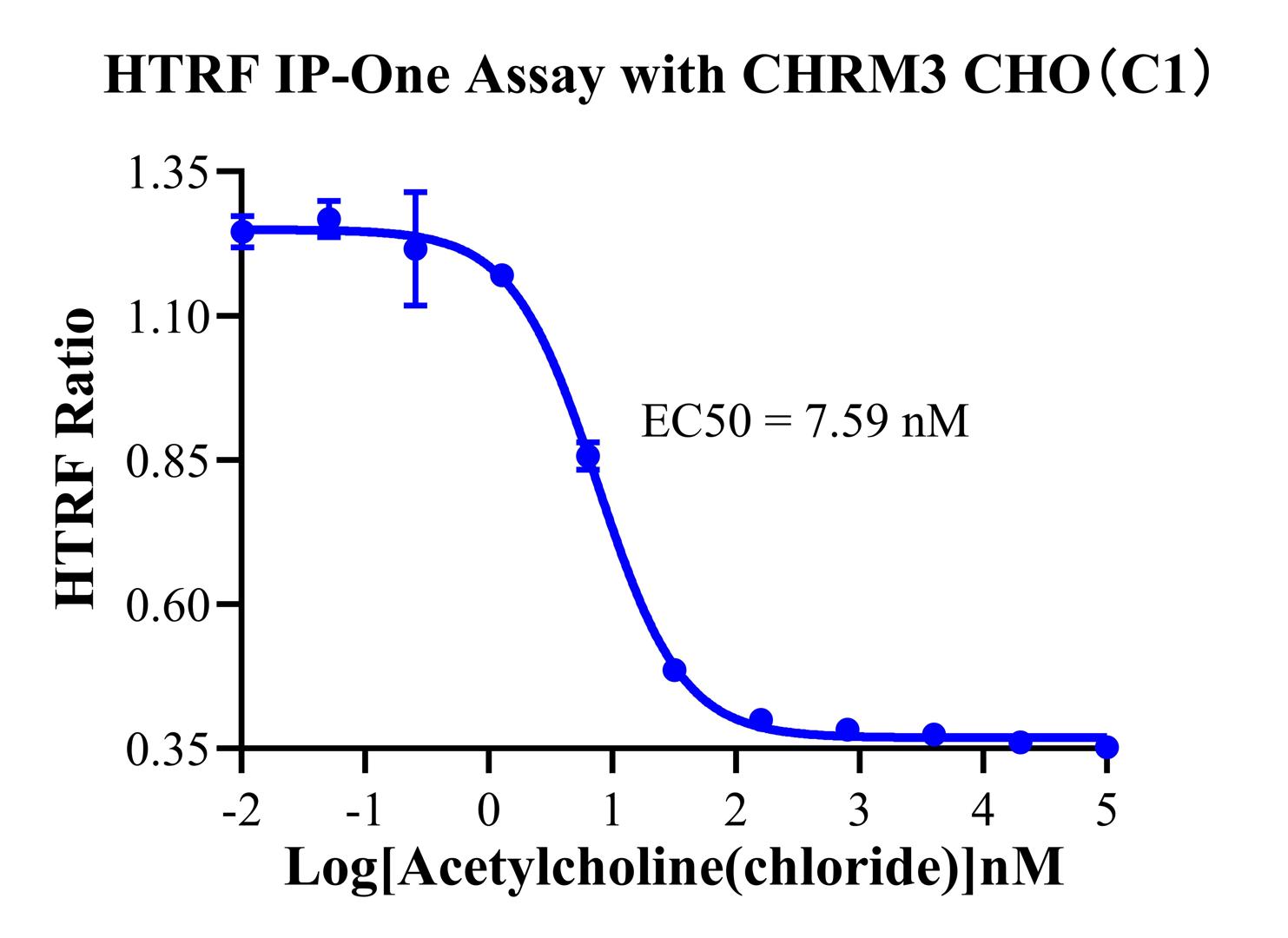
4.CHO-K1 Human CHRM4 Gα15 Cell Line(RQP71528)
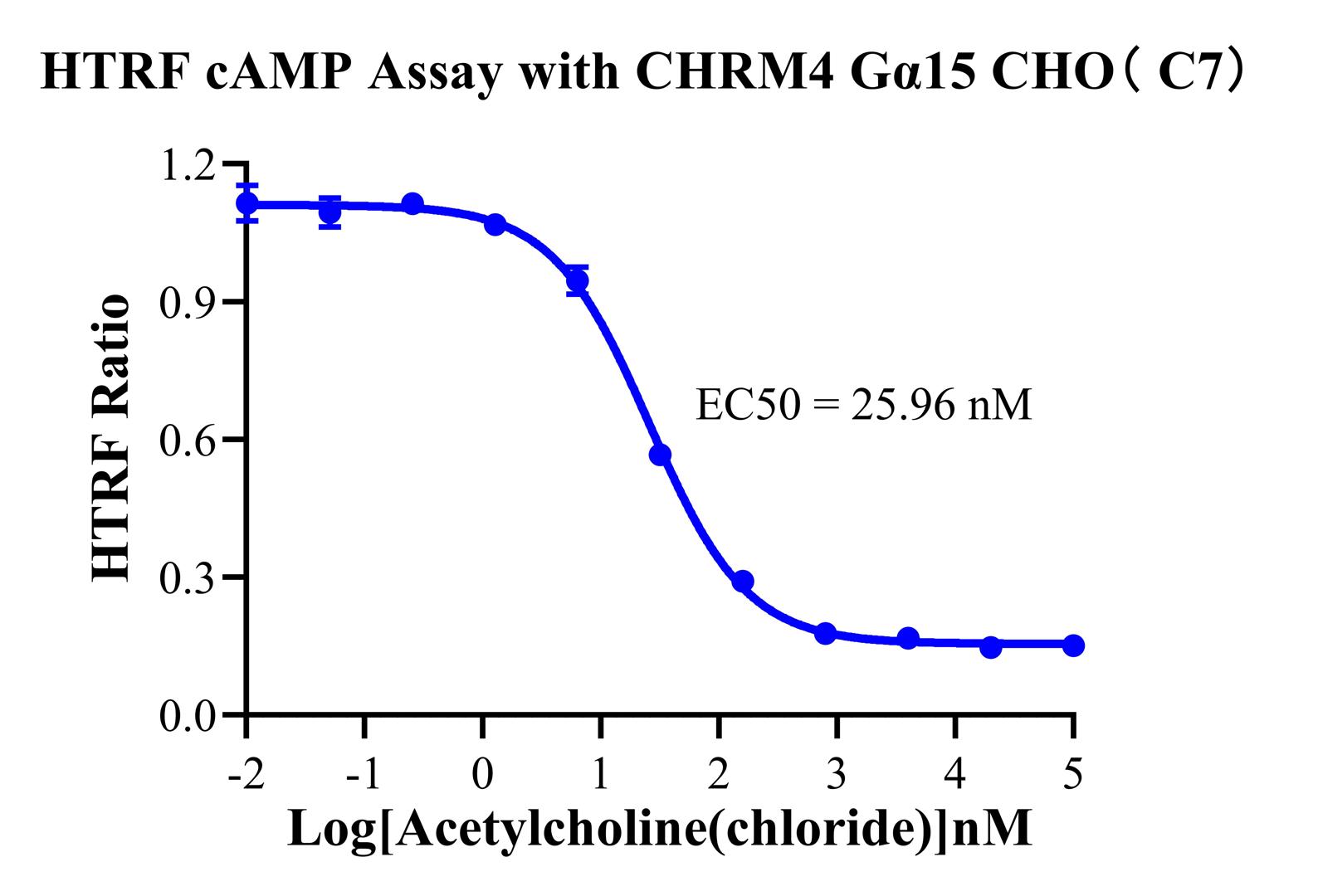
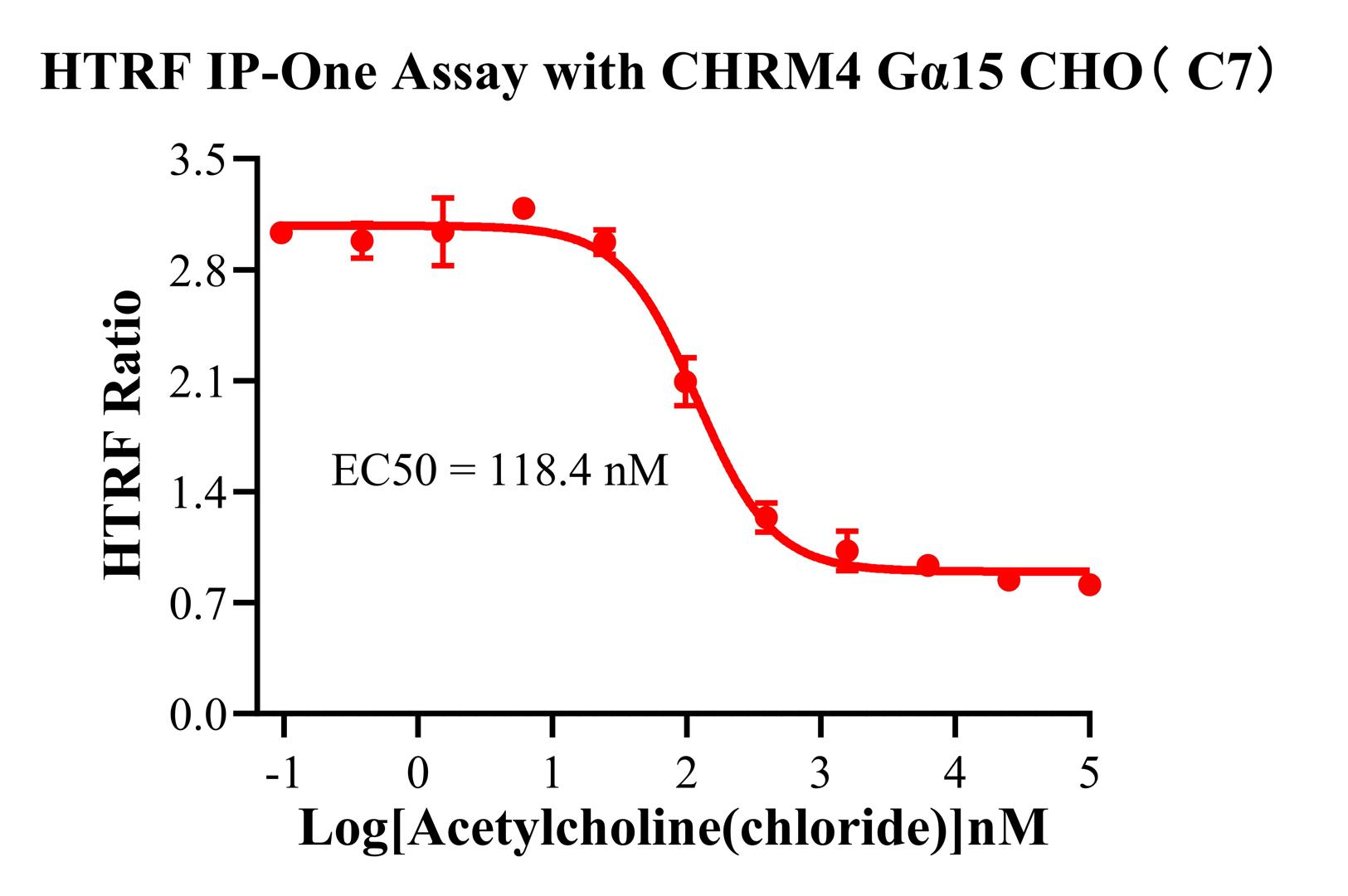
Unique Design: While wild-type M4 naturally couples with Gi (traditionally measured via cAMP inhibition), this cell line has been engineered to co-express the Gα15 protein. This modification allows M4 activation to be "re-routed" through the Gα15 pathway to activate PLCβ, making the model simultaneously compatible with IP-One assays.
Figure 9 (cAMP): Demonstrates the dose-dependent inhibition of cAMP by M4 agonists, successfully validating the functional integrity of the classic Gi pathway.
Figure 10 (IP-One): Shows that, following engineering, M4 agonists can also induce IP1 accumulation. This provides a powerful alternative for researchers who prefer not to use cAMP assays. This dual-mode validation capability is the standout feature of this specific cell model.
Summary of Core Product Advantages:
|
Advantage Dimension |
Detailed Description |
|
Full Subtype Coverage |
Offers the complete CHRM1–M5 product line to meet diverse screening needs, facilitating comprehensive subtype selectivity profiling. |
|
Platform Compatibility |
Gq-coupled subtypes utilize IP-One assays, while Gi-coupled subtypes use cAMP assays. This strictly adheres to GPCR pharmacological gold standards, ensuring highly reliable data. |
|
Unique M4 Design |
The CHRM4 Gα15 CHO cell line is compatible with both cAMP and IP-One detection modes, providing extreme flexibility to suit various laboratory equipment setups. |
|
High Sensitivity & Stability |
Each cell line undergoes monoclonal selection and rigorous functional validation (exhibiting clear dose-response curves and wide windows), ensuring exceptional batch-to-batch consistency. |
|
Ready-to-Use (RTU) |
Cell models pass strict cryopreservation QC and are ready for immediate use upon arrival, shortening R&D cycles by 2–3 months and accelerating lead discovery. |
|
Parallel Screening Support |
Enables simultaneous screening across all five subtypes to quickly evaluate a compound's selectivity profile and avoid off-target side effects (e.g., M2-mediated cardiovascular effects). |
Conclusion
The five subtypes, CHRM1–5, constitute the core network of cholinergic signaling. Their differentiated distribution and functions provide a rich array of targets for precision medical intervention. This field is currently witnessing a new surge in R&D—spanning from M1/M4 agonists for Alzheimer’s disease and M4-selective agonists for schizophrenia, to M3 antagonists for COPD and M1 antagonists for multiple sclerosis.
With a deep understanding of GPCR drug screening, ReqBio has launched a suite of high-quality cell models that offer full subtype coverage and dual-detection platforms. We are committed to empowering the global discovery and translation of innovative medicines.
news recommendation
NFAT Signaling Pathway: A Critical Target for Drug R&D and Cell Models
TSHR Cell Model for Thyroid Cancer Drug Screening | GPCR Assay Platform-Reqbio
We Are Pleased to Announce: Global Commercial Licensing Rights for Jurkat E6.1, CHO-K1, and HEK293 Cell Lines Officially Secured.
Explore



